Nitric oxide regulates angiogenesis through a functional switch involving thrombospondin-1
- PMID: 16141331
- PMCID: PMC1201580
- DOI: 10.1073/pnas.0502979102
Nitric oxide regulates angiogenesis through a functional switch involving thrombospondin-1
Abstract
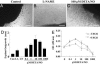
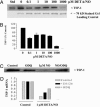
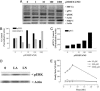
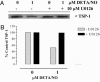
Nitric oxide (NO) donors have been shown to stimulate and inhibit the proliferation, migration, and differentiation of endothelial cells in vitro and angiogenesis in vivo. Recently, we have shown distinct thresholds for NO to regulate p53-Ser-15P, phosphorylated extracellular signal-regulated kinase (pERK), and hypoxia inducible factor 1alpha in tumor cells. Because these signaling pathways also promote the growth and survival of endothelial cells, we examined their roles in angiogenic responses of venous endothelial cells and vascular outgrowth of muscle explants elicited by NO. An additional protein involved in the regulation of angiogenesis is thrombospondin-1 (TSP1), a matricellular glycoprotein known to influence adhesion, migration, and proliferation of endothelial cells. Here we demonstrate a triphasic regulation of TSP1 mediated by a slow and prolonged release of NO that depends on ERK phosphorylation. Under conditions of 5% serum, a 24-h exposure of NO donor (0.1-1,000 microM) mediated a triphasic response in the expression of TSP1 protein: decreasing at 0.1 microM, rebounding at 100 microM, and decreasing again at 1,000 microM. Under the same conditions, we observed a dose-dependent increase in P53 phosphorylation and inverse biphasic responses of pERK and mitogen-activated protein kinase phosphatase-1. Both the growth-stimulating activity of low-dose NO for endothelial cells and suppression of TSP1 expression were ERK-dependent. Conversely, exogenous TSP1 suppressed NO-mediated pERK. These results suggest that dose-dependent positive- and negative-feedback loops exist between NO and TSP1. Limiting TSP1 expression by positive feedback through the ERK mitogen-activated protein kinase pathway may facilitate switching to a proangiogenic state at low doses of NO.
Figures

Similar articles
-
Thrombospondin-1 inhibits endothelial cell responses to nitric oxide in a cGMP-dependent manner.Proc Natl Acad Sci U S A. 2005 Sep 13;102(37):13141-6. doi: 10.1073/pnas.0502977102. Epub 2005 Sep 6. Proc Natl Acad Sci U S A. 2005. PMID: 16150726 Free PMC article.
-
The antiproliferative effect of sildenafil on pulmonary artery smooth muscle cells is mediated via upregulation of mitogen-activated protein kinase phosphatase-1 and degradation of extracellular signal-regulated kinase 1/2 phosphorylation.Anesth Analg. 2007 Oct;105(4):1034-41, table of contents. doi: 10.1213/01.ane.0000278736.81133.26. Anesth Analg. 2007. PMID: 17898384
-
Beta 1 integrin- and proteoglycan-mediated stimulation of T lymphoma cell adhesion and mitogen-activated protein kinase signaling by thrombospondin-1 and thrombospondin-1 peptides.J Immunol. 1999 Oct 1;163(7):3621-8. J Immunol. 1999. PMID: 10490955
-
Thrombospondin-1, PECAM-1, and regulation of angiogenesis.Histol Histopathol. 1999 Jan;14(1):285-94. doi: 10.14670/HH-14.285. Histol Histopathol. 1999. PMID: 9987673 Review.
-
Nitric oxide and its gatekeeper thrombospondin-1 in tumor angiogenesis.Clin Cancer Res. 2007 Feb 1;13(3):795-8. doi: 10.1158/1078-0432.CCR-06-1758. Clin Cancer Res. 2007. PMID: 17289869 Review.
Cited by
-
Improving the Quality Characteristics and Shelf Life of Meat and Growth Performance in Goose Fed Diets Supplemented with Vitamin E.Foods. 2020 Jun 17;9(6):798. doi: 10.3390/foods9060798. Foods. 2020. PMID: 32560498 Free PMC article.
-
Years of endurance exercise training remodel abdominal subcutaneous adipose tissue in adults with overweight or obesity.Nat Metab. 2024 Sep;6(9):1819-1836. doi: 10.1038/s42255-024-01103-x. Epub 2024 Sep 10. Nat Metab. 2024. PMID: 39256590
-
Oxidative stress, cancer, and sleep deprivation: is there a logical link in this association?Sleep Breath. 2013 Sep;17(3):905-10. doi: 10.1007/s11325-012-0797-9. Epub 2013 Feb 1. Sleep Breath. 2013. PMID: 23371889 Review.
-
Partial portal vein ligation plus thioacetamide: a method to obtain a new model of cirrhosis and chronic portal hypertension in the rat.J Gastrointest Surg. 2007 Feb;11(2):187-94. doi: 10.1007/s11605-006-0063-1. J Gastrointest Surg. 2007. PMID: 17390171
-
Antitumor and antiangiogenic effects of Tonantzitlolone B, an uncommon diterpene from Stillingia loranthacea.Naunyn Schmiedebergs Arch Pharmacol. 2022 Feb;395(2):267-274. doi: 10.1007/s00210-021-02185-0. Epub 2021 Dec 2. Naunyn Schmiedebergs Arch Pharmacol. 2022. PMID: 34854946
References
-
- Blaise, G. A., Gauvin, D., Gangal, M. & Authier, S. (2005) Toxicology 208, 177–192. - PubMed
-
- Dachs, G. U. & Tozer, G. M. (2000) Eur. J. Cancer 36, 1649–1660. - PubMed
-
- Wink, D. A., Vodovotz, Y., Laval, J., Laval, F., Dewhirst, M. W. & Mitchell, J. B. (1998) Carcinogenesis 19, 711–721. - PubMed
-
- Hofseth, L. J., Perwez Hussain, S., Wogan, G. N. & Harris, C. C. (2003) Free Radical Biol. Med. 34, 955–968. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

