Phase I study of continuous mitomycin-C infusion in concomitant radiochemotherapy of primary inoperable advanced head and neck cancer
- PMID: 16142489
- PMCID: PMC12161199
- DOI: 10.1007/s00432-005-0028-x
Phase I study of continuous mitomycin-C infusion in concomitant radiochemotherapy of primary inoperable advanced head and neck cancer
Abstract
Purpose: A phase I trial to evaluate the maximum tolerated dose (MTD) of continuous mitomycin-C infusion in radiochemotherapy of inoperable HNSCC.
Methods: Twenty-one patients were treated with 70 Gy (2 Gy/day) and simultaneous chemotherapy (5-FU 600 mg/m2/day and MMC, both as continuous infusion on days 1-5 and 36-40. The MMC dose was dependent on dose escalation levels I-IV: 2/2.6/3.2/4 mg/m2/day.
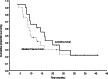
Results: Dose limiting toxicity (DLT) (grade 3 mucositis and/or dysphagia) occurred at dose level IV (MMC 4 mg/m2/day). Accordingly, dose escalation level III (MMC 3.2 mg/m2/day) was set as the MTD. One and 2-year survival rate: 66.7 and 29.5%, disease free survival: 47.6 and 22.8%, respectively.
Conclusions: Our study demonstrates that continuous infusion of 5-FU/MMC can be safely administered at a MMC dose of 3.21 mg/m2/day on days 1-5 and 36-40 in concomitant radiochemotherapy. A phase II study should be initiated to establish the role of this regimen in the treatment of head and neck cancer.
Similar articles
-
Impact of tumor control and presence of visible necrosis in head and neck cancer patients treated with radiotherapy or radiochemotherapy.J Cancer Res Clin Oncol. 2005 Nov;131(11):758-64. doi: 10.1007/s00432-005-0018-z. Epub 2005 Nov 1. J Cancer Res Clin Oncol. 2005. PMID: 16088405 Free PMC article.
-
Taxane, platinum and 5-FU prior to chemoradiotherapy benefits patients with stage IV neck node-positive head and neck cancer and a good performance status.J Cancer Res Clin Oncol. 2018 Feb;144(2):389-401. doi: 10.1007/s00432-017-2553-9. Epub 2017 Dec 8. J Cancer Res Clin Oncol. 2018. PMID: 29222650 Free PMC article.
-
Systemic therapy in the curative treatment of head and neck squamous cell cancer: a systematic review.J Otolaryngol Head Neck Surg. 2017 Apr 4;46(1):29. doi: 10.1186/s40463-017-0199-x. J Otolaryngol Head Neck Surg. 2017. PMID: 28376866 Free PMC article.
-
Gemcitabine-Based Chemoradiation in the Treatment of Locally Advanced Head and Neck Cancer: Systematic Review of Literature and Meta-Analysis.Oncologist. 2016 Jan;21(1):59-71. doi: 10.1634/theoncologist.2015-0246. Epub 2015 Dec 28. Oncologist. 2016. PMID: 26712958 Free PMC article.
-
Concurrent low-dose cisplatin and thoracic radiotherapy in patients with inoperable stage III non-small cell lung cancer: a phase II trial with special reference to the hemoglobin level as prognostic parameter.J Cancer Res Clin Oncol. 2005 Apr;131(4):261-9. doi: 10.1007/s00432-004-0633-0. Epub 2004 Dec 23. J Cancer Res Clin Oncol. 2005. PMID: 15616830 Free PMC article. Clinical Trial.
References
-
- Adam MF, Gabalski EC, Bloch DA, Oehlert JW, Brown JM, Elsaid AA, Pinto HA, Terris DJ (1999) Tissue oxygen distribution in head and neck cancer patients. Head Neck 21:146–153 - PubMed
-
- Adelstein DJ, Lavertu P, Saxton JP, Secic M, Wood BG, Wanamaker JR, Eliachar I, Strome M, Larto MA (2000) Mature results of a phase III randomized trial comparing concurrent chemoradiotherapy with radiation therapy alone in patients with stage III and IV squamous cell carcinoma of the head and neck. Cancer 88:876–883 - PubMed
-
- Adelstein DJ, Li Y, Adams GL, Wagner H Jr, Kish JA, Ensley JF, et al (2003) An intergroup phase III comparison of standard radiation therapy and two schedules of concurrent chemoradiotherapy in patients with unresectable squamous cell head and neck cancer. J Clin Oncol 21:92–98 - PubMed
-
- Brizel DM, Albers ME, Fisher SR, Scher RL, Richtsmeier WJ, Hars V, George SL, Huang AT, Prosnitz LR (1998) Hyperfractionated irradiation with or without concurrent chemotherapy for locally advanced head and neck cancer. N Engl J Med 338:1798–1804 - PubMed
-
- Browman GP, Hodson DI, Mackenzie RJ, Bestic N, Zuraw L (2001) Choosing a concomitant chemotherapy and radiotherapy regimen for squamous cell head and neck cancer: a systematic review of the published literature with subgroup analysis. Head Neck 23:579–589 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical