Comparison of three different PCR methods for quantifying human papillomavirus type 16 DNA in cervical scrape specimens
- PMID: 16145162
- PMCID: PMC1234115
- DOI: 10.1128/JCM.43.9.4868-4871.2005
Comparison of three different PCR methods for quantifying human papillomavirus type 16 DNA in cervical scrape specimens
Abstract
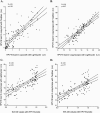
We compared real-time LightCycler and TaqMan assays and the GP5+/6+ PCR/enzyme immunoassay (EIA) to assess the human papillomavirus type 16 (HPV16) load in cervical scrape specimens. Both real-time PCR assays determined the HPV16 load in scrape specimens similarly. The level of agreement between these assays and the GP5+/6+ PCR/EIA was low (P = 0.004), suggesting that the latter method is not suited for quantifying HPV16 DNA.
Figures
Similar articles
-
Cross-sectional comparison of an automated hybrid capture 2 assay and the consensus GP5+/6+ PCR method in a population-based cervical screening program.J Clin Microbiol. 2006 Oct;44(10):3680-5. doi: 10.1128/JCM.02078-05. J Clin Microbiol. 2006. PMID: 17021097 Free PMC article.
-
Human papillomavirus quantification in urine and cervical samples by using the Mx4000 and LightCycler general real-time PCR systems.J Clin Microbiol. 2007 Mar;45(3):897-901. doi: 10.1128/JCM.02022-06. Epub 2007 Jan 17. J Clin Microbiol. 2007. PMID: 17229868 Free PMC article.
-
comparison of two commercial assays for detection of human papillomavirus (HPV) in cervical scrape specimens: validation of the Roche AMPLICOR HPV test as a means to screen for HPV genotypes associated with a higher risk of cervical disorders.J Clin Microbiol. 2005 Jun;43(6):2662-7. doi: 10.1128/JCM.43.6.2662-2667.2005. J Clin Microbiol. 2005. PMID: 15956381 Free PMC article.
-
Reliable high risk HPV DNA testing by polymerase chain reaction: an intermethod and intramethod comparison.J Clin Pathol. 1999 Jul;52(7):498-503. doi: 10.1136/jcp.52.7.498. J Clin Pathol. 1999. PMID: 10605401 Free PMC article.
-
High risk HPV load estimated by Hybrid Capture II correlates with HPV16 load measured by real-time PCR in cervical smears of HPV16-infected women.J Clin Virol. 2004 Oct;31(2):140-7. doi: 10.1016/j.jcv.2004.02.008. J Clin Virol. 2004. PMID: 15364271
Cited by
-
Can quantifying free-circulating DNA in plasma be used to identify subjects with high-grade pre-invasive endobronchial lesions?Oncol Lett. 2013 May;5(5):1591-1594. doi: 10.3892/ol.2013.1262. Epub 2013 Mar 19. Oncol Lett. 2013. PMID: 23761824 Free PMC article.
-
Integration of p16/HPV DNA Status with a 24-miRNA-Defined Molecular Phenotype Improves Clinically Relevant Stratification of Head and Neck Cancer Patients.Cancers (Basel). 2022 Jul 31;14(15):3745. doi: 10.3390/cancers14153745. Cancers (Basel). 2022. PMID: 35954409 Free PMC article.
-
Acquisition and persistence of human papillomavirus 16 (HPV-16) and HPV-18 among men with high-HPV viral load infections in a circumcision trial in Kisumu, Kenya.J Infect Dis. 2015 Mar 1;211(5):811-20. doi: 10.1093/infdis/jiu535. Epub 2014 Sep 26. J Infect Dis. 2015. PMID: 25261492 Free PMC article. Clinical Trial.
-
Comparison of GP5+/6+-PCR and SPF10-line blot assays for detection of high-risk human papillomavirus in samples from women with normal cytology results who develop grade 3 cervical intraepithelial neoplasia.J Clin Microbiol. 2008 Oct;46(10):3215-21. doi: 10.1128/JCM.00476-08. Epub 2008 Aug 6. J Clin Microbiol. 2008. PMID: 18685007 Free PMC article.
-
Cross-sectional comparison of an automated hybrid capture 2 assay and the consensus GP5+/6+ PCR method in a population-based cervical screening program.J Clin Microbiol. 2006 Oct;44(10):3680-5. doi: 10.1128/JCM.02078-05. J Clin Microbiol. 2006. PMID: 17021097 Free PMC article.
References
-
- Cuzick, J., G. Terry, L. Ho, T. Hollingworth, and M. Anderson. 1992. Human papillomavirus type 16 in cervical smears as predictor of high-grade cervical intraepithelial neoplasia [corrected]. Lancet 339:959-960. - PubMed
-
- Gravitt, P. E., R. D. Burk, A. Lorincz, R. Herrero, A. Hildesheim, M. E. Sherman, M. C. Bratti, A. C. Rodriguez, K. J. Helzlsouer, and M. Schiffman. 2003. A comparison between real-time polymerase chain reaction and hybrid capture 2 for human papillomavirus DNA quantitation. Cancer Epidemiol. Biomark. Prev. 12:477-484. - PubMed
-
- Jacobs, M. V., P. J. Snijders, A. J. van den Brule, T. J. Helmerhorst, C. J. Meijer, and J. M. Walboomers. 1997. A general primer GP5+/GP6+-mediated PCR-enzyme immunoassay method for rapid detection of 14 high-risk and 6 low-risk human papillomavirus genotypes in cervical scrapings. J. Clin. Microbiol. 35:791-795. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

