Responses of V1 neurons to two-dimensional hermite functions
- PMID: 16148274
- PMCID: PMC2927229
- DOI: 10.1152/jn.00498.2005
Responses of V1 neurons to two-dimensional hermite functions
Erratum in
- J Neurophysiol. 2006 Aug;96(2):964
Abstract
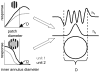
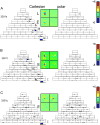
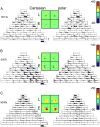
Neurons in primary visual cortex are widely considered to be oriented filters or energy detectors that perform one-dimensional feature analysis. The main deviations from this picture are generally thought to include gain controls and modulatory influences. Here we investigate receptive field (RF) properties of single neurons with localized two-dimensional stimuli, the two-dimensional Hermite functions (TDHs). TDHs can be grouped into distinct complete orthonormal bases that are matched in contrast energy, spatial extent, and spatial frequency content but differ in two-dimensional form, and thus can be used to probe spatially specific nonlinearities. Here we use two such bases: Cartesian TDHs, which resemble vignetted gratings and checkerboards, and polar TDHs, which resemble vignetted annuli and dartboards. Of 63 isolated units, 51 responded to TDH stimuli. In 37/51 units, we found significant differences in overall response size (21/51) or apparent RF shape (28/51) that depended on which basis set was used. Because of the properties of the TDH stimuli, these findings are inconsistent with simple feedforward nonlinearities and with many variants of energy models. Rather, they imply the presence of nonlinearities that are not local in either space or spatial frequency. Units showing these differences were present to a similar degree in cat and monkey, in simple and complex cells, and in supragranular, infragranular, and granular layers. We thus find a widely distributed neurophysiological substrate for two-dimensional spatial analysis at the earliest stages of cortical processing. Moreover, the population pattern of tuning to TDH functions suggests that V1 neurons sample not only orientations, but a larger space of two-dimensional form, in an even-handed manner.
Figures







Similar articles
-
Laminar and orientation-dependent characteristics of spatial nonlinearities: implications for the computational architecture of visual cortex.J Neurophysiol. 2009 Dec;102(6):3414-32. doi: 10.1152/jn.00086.2009. Epub 2009 Oct 7. J Neurophysiol. 2009. PMID: 19812295 Free PMC article.
-
Responses of striate cortex cells to grating and checkerboard patterns.J Physiol. 1979 Jun;291:483-505. doi: 10.1113/jphysiol.1979.sp012827. J Physiol. 1979. PMID: 113531 Free PMC article.
-
Relationship between contrast adaptation and orientation tuning in V1 and V2 of cat visual cortex.J Neurophysiol. 2006 Jan;95(1):271-83. doi: 10.1152/jn.00871.2005. Epub 2005 Sep 28. J Neurophysiol. 2006. PMID: 16192327
-
Population encoding of spatial frequency, orientation, and color in macaque V1.J Neurophysiol. 1994 Nov;72(5):2151-66. doi: 10.1152/jn.1994.72.5.2151. J Neurophysiol. 1994. PMID: 7884450
-
Contribution of feedforward, lateral and feedback connections to the classical receptive field center and extra-classical receptive field surround of primate V1 neurons.Prog Brain Res. 2006;154:93-120. doi: 10.1016/S0079-6123(06)54005-1. Prog Brain Res. 2006. PMID: 17010705 Review.
Cited by
-
Linear and non-linear properties of feature selectivity in V4 neurons.Front Syst Neurosci. 2015 May 27;9:82. doi: 10.3389/fnsys.2015.00082. eCollection 2015. Front Syst Neurosci. 2015. PMID: 26074788 Free PMC article.
-
Stimulus ensemble and cortical layer determine V1 spatial receptive fields.Proc Natl Acad Sci U S A. 2009 Aug 25;106(34):14652-7. doi: 10.1073/pnas.0907406106. Epub 2009 Aug 17. Proc Natl Acad Sci U S A. 2009. PMID: 19706551 Free PMC article.
-
Complexity and diversity in sparse code priors improve receptive field characterization of Macaque V1 neurons.PLoS Comput Biol. 2021 Oct 25;17(10):e1009528. doi: 10.1371/journal.pcbi.1009528. eCollection 2021 Oct. PLoS Comput Biol. 2021. PMID: 34695120 Free PMC article.
-
Contextual modulation of V1 receptive fields depends on their spatial symmetry.J Comput Neurosci. 2009 Apr;26(2):203-18. doi: 10.1007/s10827-008-0107-5. Epub 2008 Aug 5. J Comput Neurosci. 2009. PMID: 18679785 Free PMC article.
-
Information-geometric measure of 3-neuron firing patterns characterizes scale-dependence in cortical networks.J Comput Neurosci. 2011 Feb;30(1):125-41. doi: 10.1007/s10827-010-0257-0. Epub 2010 Jul 16. J Comput Neurosci. 2011. PMID: 20635129 Free PMC article.
References
-
- Abramowitz M, Stegun IA. Handbook of Mathematical Functions. Washington, DC: U.S. Department of Commerce, National Bureau of Standards; 1964. reprint, New York: Dover, 1970.
-
- Adelson EH, Bergen JR. Spatiotemporal energy models for the perception of motion. J Opt Soc Am A. 1985;2:284–299. - PubMed
-
- Albright TD, Stoner GR. Contextual influences on visual processing. Annu Rev Neurosci. 2002;25:339–379. - PubMed
-
- Aronov D, Reich DS, Mechler F, Victor JD. Neural coding of spatial phase in V1 of the macaque monkey. J Neurophysiol. 2003;89:3304–3327. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

