Phosphorylation of PI3K/Akt and MAPK/ERK in an early entry step of enterovirus 71
- PMID: 16150462
- PMCID: PMC7094582
- DOI: 10.1016/j.lfs.2005.04.076
Phosphorylation of PI3K/Akt and MAPK/ERK in an early entry step of enterovirus 71
Abstract
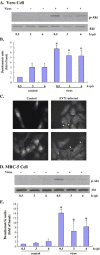
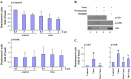
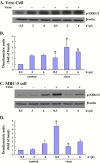
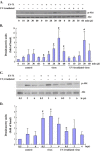
Viruses have been known to subvert the anti-apoptotic pathways of the host cell in order to delay apoptosis. However, the mechanisms utilized by enterovirus 71 (EV71) to mediate anti-apoptotic activity remained undetermined. We observed that EV71 infection induced an early activation of both phosphatidylinositol 3-kinase (PI3K)/Akt and MAPK/ERK signaling pathways. The activity of GSK3beta, a downstream target of these pathways, was negatively regulated by the activation of both MAPK/ERK and PI3K/Akt. The phosphorylation of GSK3 could be inhibited by treatment with the specific inhibitors of MAPK/ERK and PI3K/Akt. Other Akt downstream targets, BAD, caspase-9 and the Forkhead transcription factor (FKHR), were not phosphorylated during the course of infection by EV71. We further demonstrated that infection by UV-irradiated, inactivated virus triggered early Akt activation but was insufficient to trigger late Akt activation. These data suggest that with the phosphorylation of MAPK/ERK and PI3K/Akt the subsequent inactivation of GSK3beta is utilized by EV71 as a potential mechanism to delay host cell apoptosis.
Figures
Similar articles
-
Activation of PI3K/Akt pathway limits JNK-mediated apoptosis during EV71 infection.Virus Res. 2014 Nov 4;192:74-84. doi: 10.1016/j.virusres.2014.07.026. Epub 2014 Aug 10. Virus Res. 2014. PMID: 25116390
-
Indomethacin induces apoptosis in 786-O renal cell carcinoma cells by activating mitogen-activated protein kinases and AKT.Eur J Pharmacol. 2007 Jun 1;563(1-3):49-60. doi: 10.1016/j.ejphar.2007.01.071. Epub 2007 Feb 8. Eur J Pharmacol. 2007. PMID: 17341418
-
The study of traditional Chinese medical elongated-needle therapy promoting neurological recovery mechanism after spinal cord injury in rats.J Ethnopharmacol. 2016 Jul 1;187:28-41. doi: 10.1016/j.jep.2016.04.019. Epub 2016 Apr 13. J Ethnopharmacol. 2016. PMID: 27085942
-
Differential regulation of the phosphoinositide 3-kinase and MAP kinase pathways by hepatocyte growth factor vs. insulin-like growth factor-I in myogenic cells.Exp Cell Res. 2004 Jul 1;297(1):224-34. doi: 10.1016/j.yexcr.2004.03.024. Exp Cell Res. 2004. PMID: 15194438
-
Regulation of caspase-3 and -9 activation in oxidant stress to RTE by forkhead transcription factors, Bcl-2 proteins, and MAP kinases.Am J Physiol Renal Physiol. 2004 Dec;287(6):F1258-68. doi: 10.1152/ajprenal.00391.2003. Epub 2004 Aug 10. Am J Physiol Renal Physiol. 2004. PMID: 15304372
Cited by
-
Nuclear Protein Sam68 Interacts with the Enterovirus 71 Internal Ribosome Entry Site and Positively Regulates Viral Protein Translation.J Virol. 2015 Oct;89(19):10031-43. doi: 10.1128/JVI.01677-15. Epub 2015 Jul 22. J Virol. 2015. PMID: 26202240 Free PMC article.
-
Targeting cysteine-rich angiogenic inducer-61 by antibody immunotherapy suppresses growth and migration of non-small cell lung cancer.Exp Ther Med. 2018 Aug;16(2):730-738. doi: 10.3892/etm.2018.6274. Epub 2018 Jun 8. Exp Ther Med. 2018. PMID: 30116327 Free PMC article.
-
Ameobal pathogen mimivirus infects macrophages through phagocytosis.PLoS Pathog. 2008 Jun 13;4(6):e1000087. doi: 10.1371/journal.ppat.1000087. PLoS Pathog. 2008. PMID: 18551172 Free PMC article.
-
Participation of the phosphatidylinositol 3-kinase/Akt pathway in Junín virus replication in vitro.Virus Res. 2009 Oct;145(1):166-70. doi: 10.1016/j.virusres.2009.07.004. Epub 2009 Jul 10. Virus Res. 2009. PMID: 19595723 Free PMC article.
-
Suppression of astrovirus replication by an ERK1/2 inhibitor.J Virol. 2008 Aug;82(15):7475-82. doi: 10.1128/JVI.02193-07. Epub 2008 May 28. J Virol. 2008. PMID: 18508903 Free PMC article.
References
-
- Alexander J.P., Jr., Baden L., Pallansch M.A., Anderson L.J. Enterovirus 71 infections and neurologic disease—United States, 1977–1991. Journal of Infectious Diseases. 1994;169:905–908. - PubMed
-
- Brunet A., Bonni A., Zigmond M.J., Lin M.Z., Juo P., Hu L.S., Anderson M.J., Arden K.C., Blenis J., Greenberg M.E. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell. 1999;96:857–868. - PubMed
-
- Cardone M.H., Roy N., Stennicke H.R., Salvesen G.S., Franke T.F., Stanbridge E., Frisch S., Reed J.C. Regulation of cell death protease caspase-9 by phosphorylation. Science. 1998;282:1318–1321. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

