Synthesis, anti-HIV activity, and metabolic stability of new alkenyldiarylmethane HIV-1 non-nucleoside reverse transcriptase inhibitors
- PMID: 16162014
- PMCID: PMC2528834
- DOI: 10.1021/jm050452s
Synthesis, anti-HIV activity, and metabolic stability of new alkenyldiarylmethane HIV-1 non-nucleoside reverse transcriptase inhibitors
Abstract
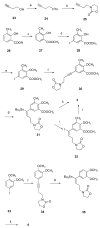
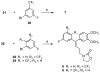
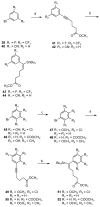
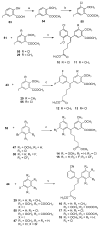
Non-nucleoside inhibitors of HIV-1 reverse transcriptase (NNRTIs) are part of the combination therapy currently used to treat HIV infection. Based on analogy with known HIV-1 NNRT inhibitors, 18 novel alkenyldiarylmethanes (ADAMs) containing 5-chloro-2-methoxyphenyl, 3-cyanophenyl, or 3-fluoro-5-trifluoromethylphenyl groups were synthesized and evaluated as HIV inhibitors. Their stabilities in rat plasma have also been investigated. Although introducing 5-chloro-2-methoxyphenyl or 3-fluoro-5-trifluoromethylphenyl groups into alkenyldiarylmethanes does not maintain the antiviral potency, the structural modification of alkenyldiarylmethanes with a 3-cyanophenyl substituent can be made without a large decrease in activity. The oxazolidinonyl group was introduced into the alkenyldiarylmethane framework and found to confer enhanced metabolic stability in rat plasma.
Figures


Similar articles
-
Design, synthesis, anti-HIV activities, and metabolic stabilities of alkenyldiarylmethane (ADAM) non-nucleoside reverse transcriptase inhibitors.J Med Chem. 2004 Jun 3;47(12):3149-62. doi: 10.1021/jm049916x. J Med Chem. 2004. PMID: 15163195
-
Synthesis and anti-HIV activity of new alkenyldiarylmethane (ADAM) non-nucleoside reverse transcriptase inhibitors (NNRTIs) incorporating benzoxazolone and benzisoxazole rings.Bioorg Med Chem. 2006 Apr 1;14(7):2366-74. doi: 10.1016/j.bmc.2005.11.014. Bioorg Med Chem. 2006. PMID: 16321539
-
Systematic evaluation of methyl ester bioisosteres in the context of developing alkenyldiarylmethanes (ADAMs) as non-nucleoside reverse transcriptase inhibitors (NNRTIs) for anti-HIV-1 chemotherapy.Bioorg Med Chem. 2016 Jul 1;24(13):3006-3022. doi: 10.1016/j.bmc.2016.05.010. Epub 2016 May 24. Bioorg Med Chem. 2016. PMID: 27234889
-
Focus on Chirality of HIV-1 Non-Nucleoside Reverse Transcriptase Inhibitors.Molecules. 2016 Feb 16;21(2):221. doi: 10.3390/molecules21020221. Molecules. 2016. PMID: 26891289 Free PMC article. Review.
-
Development of nonnucleoside HIV reverse transcriptase inhibitors.Methods Enzymol. 1996;275:440-72. doi: 10.1016/s0076-6879(96)75026-7. Methods Enzymol. 1996. PMID: 9026654 Review. No abstract available.
Cited by
-
Inhibition of tubulin polymerization by select alkenyldiarylmethanes.Bioorg Med Chem Lett. 2008 Jan 15;18(2):469-73. doi: 10.1016/j.bmcl.2007.11.114. Epub 2007 Dec 4. Bioorg Med Chem Lett. 2008. PMID: 18083556 Free PMC article.
-
Design, synthesis, and biological evaluation of conformationally constrained analogues of naphthol AS-E as inhibitors of CREB-mediated gene transcription.J Med Chem. 2012 Apr 26;55(8):4020-4. doi: 10.1021/jm300043c. Epub 2012 Apr 9. J Med Chem. 2012. PMID: 22458559 Free PMC article.
-
Investigation of the alkenyldiarylmethane non-nucleoside reverse transcriptase inhibitors as potential cAMP phosphodiesterase-4B2 inhibitors.Bioorg Med Chem Lett. 2008 Feb 15;18(4):1530-3. doi: 10.1016/j.bmcl.2007.12.015. Epub 2007 Dec 14. Bioorg Med Chem Lett. 2008. PMID: 18222088 Free PMC article.
-
Synthesis and anti-HIV activity of new metabolically stable alkenyldiarylmethane non-nucleoside reverse transcriptase inhibitors incorporating N-methoxy imidoyl halide and 1,2,4-oxadiazole systems.J Med Chem. 2007 Jul 12;50(14):3314-21. doi: 10.1021/jm070236e. Epub 2007 Jun 19. J Med Chem. 2007. PMID: 17579385 Free PMC article.
-
Synthesis of alkenyldiarylmethanes (ADAMs) containing benzo[d]isoxazole and oxazolidin-2-one rings, a new series of potent non-nucleoside HIV-1 reverse transcriptase inhibitors.Eur J Med Chem. 2009 Mar;44(3):1210-4. doi: 10.1016/j.ejmech.2008.09.013. Epub 2008 Sep 19. Eur J Med Chem. 2009. PMID: 18952324 Free PMC article.
References
-
- UNAIDS/World Health Organization. AIDS Epidemic Update, December 2004. UNAIDS/World Health Organization; Geneva: 2004.
-
- De Clercq E. Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs): Past, Present, and Future. Chem Biodiversity. 2004;1:44–64. - PubMed
-
- Esnouf R, Ren J, Ross C, Jones Y, Stammers D, Stuart D. Mechanism of Inhibition of Reverse Transcriptase by Nonnucleoside Inhibitors. Nat Struct Biol. 1995;2:303–308. - PubMed
-
- Cushman M, Golebiewski M, Buckheit RW, Jr, Graham L, Rice WG. Synthesis and Biological Evaluation of an Alkenyldiarylmethane (ADAM) which Acts as a Novel Non-nucleoside HIV-1 Reverse Transcriptase Inhibitor. Bioorg Med Chem Lett. 1995;5:2713–2716.
-
- Cushman M, Golebiewski WM, Graham L, Turpin JA, Rice WG, Fliakas-Boltz V, Buckheit RW., Jr Synthesis and Biological Evaluation of Certain Alkenyldiarylmethanes as Anti-HIV-1 Agents Which Act as Non-Nucleoside Reverse Transcriptase Inhibitors. J Med Chem. 1996;39:3217–3227. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Medical

