A role for TSLP in the development of inflammation in an asthma model
- PMID: 16172260
- PMCID: PMC2212950
- DOI: 10.1084/jem.20050199
A role for TSLP in the development of inflammation in an asthma model
Abstract
Thymic stromal lymphopoietin (TSLP) is a cytokine that promotes CD4+ T cell homeostasis. We now demonstrate that TSLP is required to mount a normal CD4+ T cell-mediated inflammatory response. TSLP acts directly on naive, but not, memory CD4+ T cells, and promotes their proliferation in response to antigen. In addition, TSLP exerts an effect indirectly through DCs to promote Th2 differentiation of CD4+ T cells. Correspondingly, TSLP receptor (TSLPR) knockout (KO) mice exhibit strong Th1 responses, with high levels of interleukin (IL)-12, interferon-gamma, and immunoglobulin (Ig) G2a, but low production of IL-4, -5, -10, -13, and IgE; moreover, CD4+ T cells from these animals proliferate less well in response to antigen. Furthermore, TSLPR KO mice fail to develop an inflammatory lung response to inhaled antigen unless supplemented with wild-type CD4+ T cells. This underscores an important role for this cytokine in the development of inflammatory and/or allergic responses in vivo.
Figures
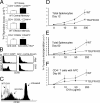


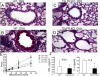


References
-
- Friend, S.L., S. Hosier, A. Nelson, D. Foxworthe, D.E. Williams, and A. Farr. 1994. A thymic stromal cell line supports in vitro development of surface IgM+ B cells and produces a novel growth factor affecting B and T lineage cells. Exp. Hematol. 22:321–328. - PubMed
-
- Park, L.S., U. Martin, K. Garka, B. Gliniak, J.P. Di Santo, W. Muller, D.A. Largaespada, N.G. Copeland, N.A. Jenkins, A.G. Farr, et al. 2000. Cloning of the murine thymic stromal lymphopoietin (TSLP) receptor: formation of a functional heteromeric complex requires interleukin 7 receptor. J. Exp. Med. 192:659–670. - PMC - PubMed
-
- Pandey, A., K. Ozaki, H. Baumann, S.D. Levin, A. Puel, A.G. Farr, S.F. Ziegler, W.J. Leonard, and H.F. Lodish. 2000. Cloning of a receptor subunit required for signaling by thymic stromal lymphopoietin. Nat. Immunol. 1:59–64. - PubMed
-
- Leonard, W.J. 2001. Cytokines and immunodeficiency diseases. Nat. Rev. Immunol. 1:200–208. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

