Effects of novel maturity-onset diabetes of the young (MODY)-associated mutations on glucokinase activity and protein stability
- PMID: 16173921
- PMCID: PMC1383698
- DOI: 10.1042/BJ20051137
Effects of novel maturity-onset diabetes of the young (MODY)-associated mutations on glucokinase activity and protein stability
Abstract
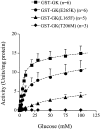
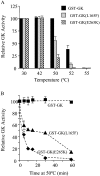
Glucokinase acts as the pancreatic glucose sensor and plays a critical role in the regulation of insulin secretion by the beta-cell. Heterozygous mutations in the glucokinase-encoding GCK gene, which result in a reduction of the enzymatic activity, cause the monogenic form of diabetes, MODY2 (maturity-onset diabetes of the young 2). We have identified and functionally characterized missense mutations in the GCK gene in diabetic families that result in protein mutations Leu165-->Phe, Glu265-->Lys and Thr206-->Met. The first two are novel GCK mutations that co-segregate with the diabetes phenotype in their respective families and are not found in more than 50 healthy control individuals. In order to measure the biochemical effects of these missense mutations on glucokinase activity, we bacterially expressed and affinity-purified islet human glucokinase proteins carrying the respective mutations and fused to GST (glutathione S-transferase). Enzymatic assays on the recombinant proteins revealed that mutations Thr206-->Met and Leu165-->Phe strongly affect the kinetic parameters of glucokinase, in agreement with the localization of both residues close to the active site of the enzyme. In contrast, mutation Glu265-->Lys, which has a weaker effect on the kinetics of glucokinase, strongly affects the protein stability, suggesting a possible structural defect of this mutant protein. Finally, none of the mutations tested appears to affect the interaction of gluco-kinase with the glucokinase regulatory protein in the yeast two-hybrid system.
Figures


Similar articles
-
Insight into the biochemical characteristics of a novel glucokinase gene mutation.Hum Genet. 2011 Mar;129(3):231-8. doi: 10.1007/s00439-010-0914-4. Epub 2010 Nov 23. Hum Genet. 2011. PMID: 21104275
-
Functional analysis of human glucokinase gene mutations causing MODY2: exploring the regulatory mechanisms of glucokinase activity.Diabetologia. 2007 Feb;50(2):325-33. doi: 10.1007/s00125-006-0542-7. Epub 2006 Dec 21. Diabetologia. 2007. PMID: 17186219
-
GCK-MODY diabetes as a protein misfolding disease: the mutation R275C promotes protein misfolding, self-association and cellular degradation.Mol Cell Endocrinol. 2014 Jan 25;382(1):55-65. doi: 10.1016/j.mce.2013.08.020. Epub 2013 Aug 31. Mol Cell Endocrinol. 2014. PMID: 24001579
-
A report of 2 new cases of MODY2 and review of the literature: implications in the search for type 2 diabetes drugs.Metabolism. 2013 Nov;62(11):1535-42. doi: 10.1016/j.metabol.2013.06.007. Epub 2013 Jul 24. Metabolism. 2013. PMID: 23890519 Review.
-
Update on mutations in glucokinase (GCK), which cause maturity-onset diabetes of the young, permanent neonatal diabetes, and hyperinsulinemic hypoglycemia.Hum Mutat. 2009 Nov;30(11):1512-26. doi: 10.1002/humu.21110. Hum Mutat. 2009. PMID: 19790256 Review.
Cited by
-
Homotropic allosteric regulation in monomeric mammalian glucokinase.Arch Biochem Biophys. 2012 Mar 15;519(2):103-11. doi: 10.1016/j.abb.2011.11.007. Epub 2011 Nov 15. Arch Biochem Biophys. 2012. PMID: 22107947 Free PMC article. Review.
-
Diabetic Kinome Inhibitors-A New Opportunity for β-Cells Restoration.Int J Mol Sci. 2021 Aug 23;22(16):9083. doi: 10.3390/ijms22169083. Int J Mol Sci. 2021. PMID: 34445786 Free PMC article. Review.
-
A vertex similarity-based framework to discover and rank orphan disease-related genes.BMC Syst Biol. 2012;6 Suppl 3(Suppl 3):S8. doi: 10.1186/1752-0509-6-S3-S8. Epub 2012 Dec 17. BMC Syst Biol. 2012. PMID: 23281592 Free PMC article.
-
Identification and functional analysis of GCK gene mutations in 12 Chinese families with hyperglycemia.J Diabetes Investig. 2019 Jul;10(4):963-971. doi: 10.1111/jdi.13001. Epub 2019 Feb 1. J Diabetes Investig. 2019. PMID: 30592380 Free PMC article.
-
Reclassification of diabetes etiology in a family with multiple diabetes phenotypes.J Clin Endocrinol Metab. 2014 Jun;99(6):E1067-71. doi: 10.1210/jc.2013-3641. Epub 2014 Feb 27. J Clin Endocrinol Metab. 2014. PMID: 24606082 Free PMC article.
References
-
- Matschinsky F. M. Glucokinase as glucose sensor and metabolic signal generator in pancreatic β-cells and hepatocytes. Diabetes. 1990;39:647–652. - PubMed
-
- Matschinsky F. M. Glucokinase, glucose homeostasis, and diabetes mellitus. Curr. Diabetes Rep. 2005;5:171–176. - PubMed
-
- Van Schaftingen E., Detheux M., Veiga da Cunha M. Short-term control of glucokinase activity: role of a regulatory protein. FASEB J. 1994;8:414–419. - PubMed
-
- Shiota C., Coffey J., Grimsby J., Grippo J. F., Magnuson M. A. Nuclear import of hepatic glucokinase depends upon glucokinase regulatory protein, whereas export is due to a nuclear export signal sequence in glucokinase. J. Biol. Chem. 1999;274:37125–37130. - PubMed
-
- Alvarez E., Roncero I., Chowen J. A., Vazquez P., Blazquez E. Evidence that glucokinase regulatory protein is expressed and interacts with glucokinase in rat brain. J. Neurochem. 2002;80:45–53. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

