NF-kappaB as a target for the prevention of graft-versus-host disease: comparative efficacy of bortezomib and PS-1145
- PMID: 16174760
- PMCID: PMC1895627
- DOI: 10.1182/blood-2005-05-1820
NF-kappaB as a target for the prevention of graft-versus-host disease: comparative efficacy of bortezomib and PS-1145
Abstract
NF-kappaB is a transcription factor that controls the expression of a number of genes important for mediating immune and inflammatory responses. In this study, we examined whether bortezomib and PS-1145, each of which inhibits NF-kappaB, could protect mice from lethal graft-versus-host disease (GVHD), which is characterized by immune activation and proinflammatory cytokine production. When administered within the first 2 days after transplantation, bortezomib and PS-1145 both protected mice from fatal GVHD, did not compromise donor engraftment, and effected marked reduction in the levels of serum cytokines that are normally increased during GVHD. Extending the course of bortezomib administration or delaying the initiation of this agent for as few as 3 days after bone marrow transplantation (BMT), however, significantly exacerbated GVHD-dependent mortality because of severe pathological damage in the colon. In contrast, prolonged administration of PS-1145, which, unlike bortezomib, is a selective inhibitor of NF-kappaB, caused no early toxicity and resulted in more complete protection than that observed with an abbreviated PS-1145 treatment schedule. These results confirm a critical role for NF-kappaB in the pathophysiology of GVHD and indicate that targeted inhibition of NF-kappaB may have a superior therapeutic index and may constitute a viable therapeutic approach to reduce GVHD severity.
Figures
 , TCD BM/spleen cells/bortezomib). Data in bar graphs are presented as the mean ± SEM. Spleen cellularity: TCD versus TCD/bortezomib, P = .53; TCD/bortezomib versus TCD/spleen/bortezomib, P = .002. Thymus cellularity: TCD versus TCD/bortezomib, P = 0.94; TCD/bortezomib versus TCD/spleen/bortezomib, P = .02.
, TCD BM/spleen cells/bortezomib). Data in bar graphs are presented as the mean ± SEM. Spleen cellularity: TCD versus TCD/bortezomib, P = .53; TCD/bortezomib versus TCD/spleen/bortezomib, P = .002. Thymus cellularity: TCD versus TCD/bortezomib, P = 0.94; TCD/bortezomib versus TCD/spleen/bortezomib, P = .02.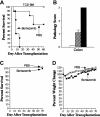
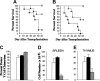
 , n = 8) or bortezomib (▪, n = 7). In panels C and D, lethally irradiated B6 mice underwent transplantation with 10 × 106 B6.SJL BM cells and then were treated with twice-weekly PBS (□, n = 10) or bortezomib (▪, n = 10) (1 mg/kg/dose) beginning 3 to 4 days after BMT. Survival is shown in panel C, and panel D shows mean percentages of pretransplantation weight loss/gain. Data are cumulative results from 2 independent experiments for each strain combination (allogeneic and syngeneic).
, n = 8) or bortezomib (▪, n = 7). In panels C and D, lethally irradiated B6 mice underwent transplantation with 10 × 106 B6.SJL BM cells and then were treated with twice-weekly PBS (□, n = 10) or bortezomib (▪, n = 10) (1 mg/kg/dose) beginning 3 to 4 days after BMT. Survival is shown in panel C, and panel D shows mean percentages of pretransplantation weight loss/gain. Data are cumulative results from 2 independent experiments for each strain combination (allogeneic and syngeneic).
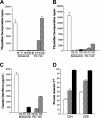
 ) for 5 days. Cells were pulsed with 3H-Tdr for the last 18 hours of culture, and the percentages of incorporated radioactivity were determined. (B-C) B10.BR T cells (1 × 105/well) were cultured with B6 CD11c+ dendritic cells (5 × 104) alone or with bortezomib (0.32 μM) or PS-1145 (8 μM) for 5 days. Bortezomib and PS-1145 were added to microwells on days 0, 1, or 2 after culture initiation. Cells were pulsed with 3H-Tdr for the last 18 hours of culture and the percentages of incorporated radioactivity were determined (B); in separate experiments, supernatants were collected and tested for the presence of IFN-γ (C). Data are presented as the mean ± SEM from triplicate control wells. Data shown are from 1 of 3 experiments that produced similar results. (D) B10.BR T cells were cultured with B6 CD11c+ dendritic cells alone or with bortezomib (0.32 μM) or PS-1145 (8 μM). Cells were harvested and pooled from triplicate microwells after 3 or 4 days and were surface stained for CD4 or CD8 and annexin V. Data are presented as the mean ± SEM. Data shown are cumulative results of 4 experiments.
) for 5 days. Cells were pulsed with 3H-Tdr for the last 18 hours of culture, and the percentages of incorporated radioactivity were determined. (B-C) B10.BR T cells (1 × 105/well) were cultured with B6 CD11c+ dendritic cells (5 × 104) alone or with bortezomib (0.32 μM) or PS-1145 (8 μM) for 5 days. Bortezomib and PS-1145 were added to microwells on days 0, 1, or 2 after culture initiation. Cells were pulsed with 3H-Tdr for the last 18 hours of culture and the percentages of incorporated radioactivity were determined (B); in separate experiments, supernatants were collected and tested for the presence of IFN-γ (C). Data are presented as the mean ± SEM from triplicate control wells. Data shown are from 1 of 3 experiments that produced similar results. (D) B10.BR T cells were cultured with B6 CD11c+ dendritic cells alone or with bortezomib (0.32 μM) or PS-1145 (8 μM). Cells were harvested and pooled from triplicate microwells after 3 or 4 days and were surface stained for CD4 or CD8 and annexin V. Data are presented as the mean ± SEM. Data shown are cumulative results of 4 experiments.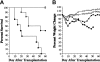
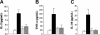
 ) or PS-1145 (50 mg/kg, days 0-2) (
) or PS-1145 (50 mg/kg, days 0-2) ( ). Mice in all groups were bled on day 7 after BMT and were analyzed for the specified cytokines. Data are cumulative results from 2 independent experiments with 5 to 7 mice per group. Data are presented as the mean ± SEM. IL-2: bortezomib or PS-1145 versus GVHD control, P < .005. TNF-α: bortezomib or PS-1145 versus GVHD control, P = .02. IL-10: bortezomib or PS-1145 versus GVHD control, P < .005.
). Mice in all groups were bled on day 7 after BMT and were analyzed for the specified cytokines. Data are cumulative results from 2 independent experiments with 5 to 7 mice per group. Data are presented as the mean ± SEM. IL-2: bortezomib or PS-1145 versus GVHD control, P < .005. TNF-α: bortezomib or PS-1145 versus GVHD control, P = .02. IL-10: bortezomib or PS-1145 versus GVHD control, P < .005.
 , DMSO; ▪, PS-1145). Data in bar graphs are presented as the mean ± SEM. Spleen cellularity: DMSO versus PS-1145, P = .006; TCD versus PS-1145, P < .001. Total splenic B cells: DMSO versus PS-1145, P = .04; TCD versus PS-1145, P < .001. Percentage donor T cells: DMSO versus PS-1145, P = .66; TCD versus PS-1145, P < .001
, DMSO; ▪, PS-1145). Data in bar graphs are presented as the mean ± SEM. Spleen cellularity: DMSO versus PS-1145, P = .006; TCD versus PS-1145, P < .001. Total splenic B cells: DMSO versus PS-1145, P = .04; TCD versus PS-1145, P < .001. Percentage donor T cells: DMSO versus PS-1145, P = .66; TCD versus PS-1145, P < .001Similar articles
-
Inhibition of acute graft-versus-host disease with retention of graft-versus-tumor effects by the proteasome inhibitor bortezomib.Proc Natl Acad Sci U S A. 2004 May 25;101(21):8120-5. doi: 10.1073/pnas.0401563101. Epub 2004 May 17. Proc Natl Acad Sci U S A. 2004. PMID: 15148407 Free PMC article.
-
Immune-modulatory effects of bortezomib in GVHD.Leuk Lymphoma. 2007 May;48(5):853-4. doi: 10.1080/10428190701297394. Leuk Lymphoma. 2007. PMID: 17487726 No abstract available.
-
Differential effects of proteasome inhibition by bortezomib on murine acute graft-versus-host disease (GVHD): delayed administration of bortezomib results in increased GVHD-dependent gastrointestinal toxicity.Blood. 2005 Nov 1;106(9):3293-9. doi: 10.1182/blood-2004-11-4526. Epub 2005 Jun 16. Blood. 2005. PMID: 15961519 Free PMC article.
-
Use of systemic proteasome inhibition as an immune-modulating agent in disease.Endocr Metab Immune Disord Drug Targets. 2007 Mar;7(1):29-34. doi: 10.2174/187153007780059397. Endocr Metab Immune Disord Drug Targets. 2007. PMID: 17346202 Review.
-
Can NF-kappaB be a target for novel and efficient anti-cancer agents?Biochem Pharmacol. 2006 Oct 30;72(9):1054-68. doi: 10.1016/j.bcp.2006.07.023. Epub 2006 Sep 14. Biochem Pharmacol. 2006. PMID: 16973133 Review.
Cited by
-
Bortezomib-based graft-versus-host disease prophylaxis in HLA-mismatched unrelated donor transplantation.J Clin Oncol. 2012 Sep 10;30(26):3202-8. doi: 10.1200/JCO.2012.42.0984. Epub 2012 Aug 6. J Clin Oncol. 2012. PMID: 22869883 Free PMC article. Clinical Trial.
-
Current and emerging strategies for the prevention of graft-versus-host disease.Nat Rev Clin Oncol. 2014 Sep;11(9):536-47. doi: 10.1038/nrclinonc.2014.102. Epub 2014 Jun 24. Nat Rev Clin Oncol. 2014. PMID: 24958183 Free PMC article. Review.
-
Sequential activation of inflammatory signaling pathways during graft-versus-host disease (GVHD): early role for STAT1 and STAT3.Cell Immunol. 2011;268(1):37-46. doi: 10.1016/j.cellimm.2011.01.008. Epub 2011 Feb 4. Cell Immunol. 2011. PMID: 21376308 Free PMC article.
-
Fasting and refeeding differentially regulate NLRP3 inflammasome activation in human subjects.J Clin Invest. 2015 Nov 3;125(12):4592-600. doi: 10.1172/JCI83260. J Clin Invest. 2015. PMID: 26529255 Free PMC article. Clinical Trial.
-
Advances in Immunosuppressive Agents Based on Signal Pathway.Front Pharmacol. 2022 May 26;13:917162. doi: 10.3389/fphar.2022.917162. eCollection 2022. Front Pharmacol. 2022. PMID: 35694243 Free PMC article. Review.
References
-
- Ferrara JLM, Deeg HJ, Burakoff S. Graft Versus Host Disease. New York, NY: Marcell Dekker.
-
- Xun CQ, Thompson JS, Jennings CD, Brown SA, Widmer MB. Effect of total body irradiation, busul-fan-cyclophosphamide, or cyclophosphamide conditioning on inflammatory cytokine release and development of acute and chronic graft versus host disease in H-2 incompatible transplanted SCID mice. Blood. 1994;83: 2360-2367. - PubMed
-
- Gonzalez M, Quezada SA, Blazar BR, Panoskaltsis-Mortari A, Rudensky AY, Noelle RJ. The balance between donor T cell anergy and suppression versus lethal graft-versus-host disease is determined by host conditioning. J Immunol. 2002;169: 5581-5589. - PubMed
-
- Reddy P. Pathophysiology of acute graft-versus-host disease. Hematol Oncol. 2003;21: 149-161. - PubMed
-
- Shlomchik WD, Couzens MS, Tang CB, et al. Prevention of graft versus host disease by inactivation of host antigen-presenting cells. Science. 1999;285: 412-415. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

