Expression and functional phenotype of mouse ERG K+ channels in the inner ear: potential role in K+ regulation in the inner ear
- PMID: 16177035
- PMCID: PMC6725506
- DOI: 10.1523/JNEUROSCI.1422-05.2005
Expression and functional phenotype of mouse ERG K+ channels in the inner ear: potential role in K+ regulation in the inner ear
Abstract
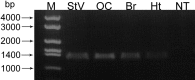
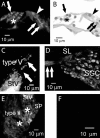
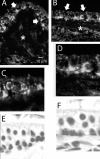
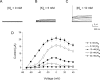
An outcome of the intricate K+ regulation in the cochlear duct is the endocochlear potential (EP), approximately 80 mV, the "battery" that runs hair-cell transduction; however, the detailed molecular mechanisms for the generation of the EP remain unclear. We provide strong evidence indicating that the intermediate cells (ICs) of the stria vascularis (StV) express outward K+ current that rectifies inwardly at positive potentials. The channel belongs to the ether-a-go-go-related gene (erg) family of K+ channels. We cloned an ERG1a channel in the mouse inner ear (MERG1a). The cellular distribution of MERG1a in the cochlea displayed the highest levels of immunoreactivity in the ICs and modest reactivity in the marginal cells as well as in several extrastrial cells (e.g., hair cells). Functional expression of the StV-specific MERG1a channel reveals a current that activates at relatively negative potentials (approximately-50 mV) and shows rapid inactivation reflected as inward rectification at depolarized potentials. The current was sensitive to the methanesulfonanilide drug E-4031 (IC50, approximately 165 nM) and the recombinant peptide rBeKm-1 (IC50, approximately 16 nM), and the single-channel conductance in symmetrical K+ was approximately 14 pS. The site of expression of MERG1a and its functional phenotype (e.g., modulation of the current by external K+ make it one of the most likely candidates for establishing the high throughput of K+ ions across ICs to generate EP. In addition, the property of the channel that produces marked K+ extrusion in increased external K+ may be important in shaping the dynamics of K+ cycling in the inner ear.
Figures






Similar articles
-
Expression and role of the ether-à-go-go-related (MERG1A) potassium-channel protein during preimplantation mouse development.Biol Reprod. 2004 Apr;70(4):1070-9. doi: 10.1095/biolreprod.103.020917. Epub 2003 Dec 10. Biol Reprod. 2004. PMID: 14668215
-
Two isoforms of the mouse ether-a-go-go-related gene coassemble to form channels with properties similar to the rapidly activating component of the cardiac delayed rectifier K+ current.Circ Res. 1997 Nov;81(5):870-8. doi: 10.1161/01.res.81.5.870. Circ Res. 1997. PMID: 9351462
-
ERG K+ currents regulate pacemaker activity in ICC.Am J Physiol Gastrointest Liver Physiol. 2003 Dec;285(6):G1249-58. doi: 10.1152/ajpgi.00149.2003. Epub 2003 Sep 4. Am J Physiol Gastrointest Liver Physiol. 2003. PMID: 12958021
-
Erg K+ channels modulate contractile activity in the bovine epididymal duct.Am J Physiol Regul Integr Comp Physiol. 2008 Mar;294(3):R895-904. doi: 10.1152/ajpregu.00521.2007. Epub 2008 Jan 9. Am J Physiol Regul Integr Comp Physiol. 2008. PMID: 18184764
-
An ATP-dependent inwardly rectifying potassium channel, KAB-2 (Kir4. 1), in cochlear stria vascularis of inner ear: its specific subcellular localization and correlation with the formation of endocochlear potential.J Neurosci. 1997 Jun 15;17(12):4711-21. doi: 10.1523/JNEUROSCI.17-12-04711.1997. J Neurosci. 1997. PMID: 9169531 Free PMC article.
Cited by
-
M-like K+ currents in type I hair cells and calyx afferent endings of the developing rat utricle.J Neurosci. 2006 Oct 4;26(40):10253-69. doi: 10.1523/JNEUROSCI.2596-06.2006. J Neurosci. 2006. PMID: 17021181 Free PMC article.
-
Dietary thyroid hormone replacement ameliorates hearing deficits in hypothyroid mice.Mamm Genome. 2007 Aug;18(8):596-608. doi: 10.1007/s00335-007-9038-0. Epub 2007 Sep 22. Mamm Genome. 2007. PMID: 17899304
-
Supporting sensory transduction: cochlear fluid homeostasis and the endocochlear potential.J Physiol. 2006 Oct 1;576(Pt 1):11-21. doi: 10.1113/jphysiol.2006.112888. Epub 2006 Jul 20. J Physiol. 2006. PMID: 16857713 Free PMC article. Review.
-
Mouse ERG K(+) channel clones reveal differences in protein trafficking and function.J Am Heart Assoc. 2014 Dec 11;3(6):e001491. doi: 10.1161/JAHA.114.001491. J Am Heart Assoc. 2014. PMID: 25497881 Free PMC article.
-
HERG1 channelopathies.Pflugers Arch. 2010 Jul;460(2):265-76. doi: 10.1007/s00424-009-0758-8. Epub 2009 Nov 22. Pflugers Arch. 2010. PMID: 20544339 Free PMC article. Review.
References
-
- Abbott GW, Sesti F, Splawski I, Buck ME, Lehmann MH, Timothy KW, Keating MT, Goldstein SA (1999) MiRP1 forms IKr potassium channels with HERG and is associated with cardiac arrhythmia. Cell 97: 175–187. - PubMed
-
- Arcangeli A, Rosati B, Cherubini A, Crociani O, Fontana L, Ziller C, Wanke E, Olivotto M (1997) HERG- and IRK-like inward rectifier currents are sequentially expressed during neuronal development of neural crest cells and their derivatives. Eur J Neurosci 9: 2596–2604. - PubMed
-
- Carlisle L, Steel K, Forge A (1990) Endocochlear potential generation is associated with intercellular communication in the stria vascularis: structural analysis in the viable dominant spotting mouse mutant. Cell Tissue Res 262: 329–337. - PubMed
-
- Casimiro MC, Knollmann BC, Yamoah EN, Nie L, Vary JC, Surenko S, Greene AE, Grinberg A, Huang SP, Ebert SN, Pfeifer K (2004) Targeted point mutagenesis of mouse Kcnq1: phenotypic analysis of mice with point mutations that cause Romano-Ward syndrome in humans. Genomics 84: 555–564. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
