Comparative opsonic and protective activities of Staphylococcus aureus conjugate vaccines containing native or deacetylated Staphylococcal Poly-N-acetyl-beta-(1-6)-glucosamine
- PMID: 16177353
- PMCID: PMC1230901
- DOI: 10.1128/IAI.73.10.6752-6762.2005
Comparative opsonic and protective activities of Staphylococcus aureus conjugate vaccines containing native or deacetylated Staphylococcal Poly-N-acetyl-beta-(1-6)-glucosamine
Erratum in
- Infect Immun. 2005 Nov;73(11):7789
Abstract
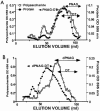
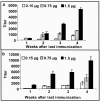
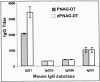
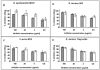
Staphylococcus aureus and Staphylococcus epidermidis both synthesize the surface polysaccharide poly-N-acetyl-beta-(1-6)-glucosamine (PNAG), which is produced in vitro with a high level (>90%) of the amino groups substituted by acetate. Here, we examined the role of the acetate substituents of PNAG in generating opsonic and protective antibodies. PNAG and a deacetylated form of the antigen (dPNAG; 15% acetylation) were conjugated to the carrier protein diphtheria toxoid (DT) and used to immunize animals. Mice responded in a dose-dependent fashion to both conjugate vaccines, with maximum antibody titers observed at the highest dose and 4 weeks after the last of three weekly immunizations. PNAG-DT and dPNAG-DT vaccines were also very immunogenic in rabbits. Antibodies raised to the conjugate vaccines in rabbits mediated the opsonic killing of various staphylococcal strains, but the specificity of the opsonic killing was primarily to dPNAG, as this antigen inhibited the killing of S. aureus strains by both PNAG- and dPNAG-specific antibodies. Passive immunization of mice with anti-dPNAG-DT rabbit sera showed significant levels of clearance of S. aureus from the blood (54 to 91%) compared to control mice immunized with normal rabbit sera, whereas PNAG-specific antibodies were ineffective at clearing S. aureus. Passive immunization of mice with a goat antiserum raised to the dPNAG-DT vaccine protected against a lethal dose of three different S. aureus strains. Overall, these data show that immunization of animals with a conjugate vaccine of dPNAG elicit antibodies that mediated opsonic killing and protected against S. aureus infection, including capsular polysaccharide types 5 and 8 and an untypable strain.
Figures



References
-
- Abraham, J., C. Mansour, E. Veledar, B. Khan, and S. Lerakis. 2004. Staphylococcus aureus bacteremia and endocarditis: the Grady Memorial Hospital experience with methicillin-sensitive S aureus and methicillin-resistant S aureus bacteremia. Am. Heart J. 147:536-539. - PubMed
-
- Baba, T., F. Takeuchi, M. Kuroda, H. Yuzawa, K. Aoki, A. Oguchi, Y. Nagai, N. Iwama, K. Asano, T. Naimi, H. Kuroda, L. Cui, K. Yamamoto, and K. Hiramatsu. 2002. Genome and virulence determinants of high virulence community-acquired MRSA. Lancet 359:1819-1827. - PubMed
-
- Bogaert, D., P. W. Hermans, P. V. Adrian, H. C. Rumke, and R. de Groot. 2004. Pneumococcal vaccines: an update on current strategies. Vaccine 22:2209-2220. - PubMed
-
- Bradford, M. M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248-254. - PubMed
-
- Bystricky, S., E. Machova, P. Bartek, N. Kolarova, and G. Kogan. 2000. Conjugation of yeast mannans with protein employing cyanopyridinium agent (CDAP)—an effective route of antifungal vaccine preparation. Glycoconj. J. 17:677-680. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

