Akt phosphorylates acinus and inhibits its proteolytic cleavage, preventing chromatin condensation
- PMID: 16177823
- PMCID: PMC1276706
- DOI: 10.1038/sj.emboj.7600823
Akt phosphorylates acinus and inhibits its proteolytic cleavage, preventing chromatin condensation
Abstract
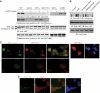
Akt promotes cell survival by phosphorylating and inhibiting components of the intrinsic cell death machinery. Akt translocates into the nucleus upon exposure of cells to survival factors, but little is known about its functions in the nucleus. Here, we show that acinus, a nuclear factor required for apoptotic chromatin condensation, is a direct target of Akt. We demonstrate that Akt phosphorylation of acinus on serine 422 and 573 results in its resistance to caspase cleavage in the nucleus and the inhibition of acinus-dependent chromatin condensation. Abolishing acinus phosphorylation by Akt through mutagenesis accelerates its proteolytic degradation and chromatin condensation. Acinus S422, 573D, a mutant mimicking phosphorylation, resists against apoptotic cleavage and prevents chromatin condensation. Knocking down of acinus substantially decreases chromatin condensation, and depletion of Akt provokes the apoptotic cleavage of acinus. Thus, Akt inhibits chromatin condensation during apoptosis by phosphorylating acinus in the nucleus, revealing a specific mechanism by which nuclear Akt promotes cell survival.
Figures





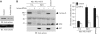
Similar articles
-
Akt phosphorylation of zyxin mediates its interaction with acinus-S and prevents acinus-triggered chromatin condensation.Cell Death Differ. 2007 Sep;14(9):1688-99. doi: 10.1038/sj.cdd.4402179. Epub 2007 Jun 15. Cell Death Differ. 2007. PMID: 17572661
-
Loss of Acinus inhibits oligonucleosomal DNA fragmentation but not chromatin condensation during apoptosis.J Biol Chem. 2006 May 5;281(18):12475-84. doi: 10.1074/jbc.M509859200. Epub 2006 Mar 14. J Biol Chem. 2006. PMID: 16537548
-
Acinus-provoked protein kinase C delta isoform activation is essential for apoptotic chromatin condensation.Cell Death Differ. 2007 Dec;14(12):2035-46. doi: 10.1038/sj.cdd.4402214. Epub 2007 Aug 24. Cell Death Differ. 2007. PMID: 17721436
-
The emerging multiple roles of nuclear Akt.Biochim Biophys Acta. 2012 Dec;1823(12):2168-78. doi: 10.1016/j.bbamcr.2012.08.017. Epub 2012 Aug 31. Biochim Biophys Acta. 2012. PMID: 22960641 Review.
-
PIKE/nuclear PI 3-kinase signaling in preventing programmed cell death.J Cell Biochem. 2005 Oct 15;96(3):463-72. doi: 10.1002/jcb.20549. J Cell Biochem. 2005. PMID: 16088938 Review.
Cited by
-
Crystal structure of human Acinus RNA recognition motif domain.PeerJ. 2018 Jul 4;6:e5163. doi: 10.7717/peerj.5163. eCollection 2018. PeerJ. 2018. PMID: 30042883 Free PMC article.
-
An unbiased proteomic screen reveals caspase cleavage is positively and negatively regulated by substrate phosphorylation.Mol Cell Proteomics. 2014 May;13(5):1184-97. doi: 10.1074/mcp.M113.037374. Epub 2014 Feb 20. Mol Cell Proteomics. 2014. PMID: 24556848 Free PMC article.
-
Interaction of Akt-phosphorylated SRPK2 with 14-3-3 mediates cell cycle and cell death in neurons.J Biol Chem. 2009 Sep 4;284(36):24512-25. doi: 10.1074/jbc.M109.026237. Epub 2009 Jul 10. J Biol Chem. 2009. PMID: 19592491 Free PMC article.
-
The androgen receptor-lncRNASAT1-AKT-p15 axis mediates androgen-induced cellular senescence in prostate cancer cells.Oncogene. 2022 Feb;41(7):943-959. doi: 10.1038/s41388-021-02060-5. Epub 2021 Oct 19. Oncogene. 2022. PMID: 34667276 Free PMC article.
-
Acinus-S' represses retinoic acid receptor (RAR)-regulated gene expression through interaction with the B domains of RARs.Mol Cell Biol. 2008 Apr;28(8):2549-58. doi: 10.1128/MCB.01199-07. Epub 2008 Feb 4. Mol Cell Biol. 2008. PMID: 18250153 Free PMC article.
References
-
- Ahmed NN, Franke TF, Bellacosa A, Datta K, Gonzalez-Portal ME, Taguchi T, Testa JR, Tsichlis PN (1993) The proteins encoded by c-akt and v-akt differ in post-translational modification, subcellular localization and oncogenic potential. Oncogene 8: 1957–1963 - PubMed
-
- Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS, Anderson MJ, Arden KC, Blenis J, Greenberg ME (1999) Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96: 857–868 - PubMed
-
- Brunet A, Datta SR, Greenberg ME (2001) Transcription-dependent and -independent control of neuronal survival by the PI3K–Akt signaling pathway. Curr Opin Neurobiol 11: 297–305 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

