Reduced iC3b-mediated phagocytotic capacity of pulmonary neutrophils in cystic fibrosis
- PMID: 16178858
- PMCID: PMC1809487
- DOI: 10.1111/j.1365-2249.2005.02893.x
Reduced iC3b-mediated phagocytotic capacity of pulmonary neutrophils in cystic fibrosis
Abstract
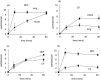
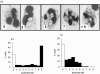
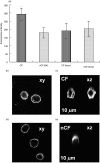
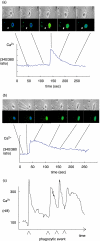
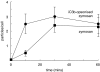
Cystic fibrosis (CF) is characterized by a neutrophil-dominated chronic inflammation of the airways with persistent infections. In order to investigate whether neutrophils contribute to an inadequacy in the pulmonary defence mechanism, the phagocytic activity of pulmonary and peripheral blood neutrophils from CF and non-CF respiratory patients were compared. Neutrophils were isolated from both the blood and bronchoalveolar lavage fluid of 21 patients with CF (12 male, 9 female; mean age 7.5 years, range 0.25-16.4 years) and 17 non-CF subjects (9 male, 8 female; mean age 5.4 years, range 0.2-13.1 years). The ex vivo phagocytic rate of normal pulmonary neutrophils to internalize zymosan particles opsonized with iC3b was faster than that of circulating neutrophils (P < 0.05), but the maximum capacity (9 particles/cell) was similar. In contrast, pulmonary neutrophils from patients with CF had a lower phagocytic capacity than circulating neutrophils either from the same patients or from normal subjects. This deficiency could not be attributed to (i) the cell surface density of CR3 (CD18/CD11b) receptors, which were not significantly different between the other groups (ii) the signalling ability of the CR3 receptors, using cytosolic free Ca(2+) signalling as the receptor activity read-out or (iii) a decrease in cellular ATP concentration. As CFTR was not detectable on neutrophils from any source by either histochemistry or Western blotting, it was concluded that the reduced phagocytic capacity was not the direct result of a CFTR mutation, but was attributed to a failure of neutrophil phagocytic priming during translocation into the CF lung.
Figures
Similar articles
-
The beta-glucan-binding lectin site of mouse CR3 (CD11b/CD18) and its function in generating a primed state of the receptor that mediates cytotoxic activation in response to iC3b-opsonized target cells.J Immunol. 1999 Feb 15;162(4):2281-90. J Immunol. 1999. PMID: 9973505
-
Optimal complement-mediated phagocytosis of Pseudomonas aeruginosa by monocytes is cystic fibrosis transmembrane conductance regulator-dependent.Am J Respir Cell Mol Biol. 2013 Sep;49(3):463-70. doi: 10.1165/rcmb.2012-0502OC. Am J Respir Cell Mol Biol. 2013. PMID: 23617438
-
Neutrophil adhesion molecule surface expression and responsiveness in cystic fibrosis.Am J Respir Crit Care Med. 1998 Mar;157(3 Pt 1):756-61. doi: 10.1164/ajrccm.157.3.9704008. Am J Respir Crit Care Med. 1998. PMID: 9517587
-
The cystic fibrosis neutrophil: a specialized yet potentially defective cell.Arch Immunol Ther Exp (Warsz). 2011 Apr;59(2):97-112. doi: 10.1007/s00005-011-0113-6. Epub 2011 Feb 11. Arch Immunol Ther Exp (Warsz). 2011. PMID: 21311988 Review.
-
Neutrophils in cystic fibrosis.Biol Chem. 2016 Jun 1;397(6):485-96. doi: 10.1515/hsz-2015-0271. Biol Chem. 2016. PMID: 26854289 Review.
Cited by
-
Pathogenesis, imaging and clinical characteristics of CF and non-CF bronchiectasis.BMC Pulm Med. 2018 May 22;18(1):79. doi: 10.1186/s12890-018-0630-8. BMC Pulm Med. 2018. PMID: 29788954 Free PMC article. Review.
-
Anti-Pseudomonas aeruginosa IgY antibodies promote bacterial opsonization and augment the phagocytic activity of polymorphonuclear neutrophils.Hum Vaccin Immunother. 2016 Jul 2;12(7):1690-9. doi: 10.1080/21645515.2016.1145848. Epub 2016 Feb 22. Hum Vaccin Immunother. 2016. PMID: 26901841 Free PMC article.
-
Inhibiting CFTR through inh-172 in primary neutrophils reveals CFTR-specific functional defects.Sci Rep. 2024 Dec 28;14(1):31237. doi: 10.1038/s41598-024-82535-z. Sci Rep. 2024. PMID: 39732786 Free PMC article.
-
Inhaled hypertonic saline for cystic fibrosis: Reviewing the potential evidence for modulation of neutrophil signalling and function.World J Crit Care Med. 2015 Aug 4;4(3):179-91. doi: 10.5492/wjccm.v4.i3.179. eCollection 2015 Aug 4. World J Crit Care Med. 2015. PMID: 26261770 Free PMC article. Review.
-
Pf Bacteriophage and Their Impact on Pseudomonas Virulence, Mammalian Immunity, and Chronic Infections.Front Immunol. 2020 Feb 21;11:244. doi: 10.3389/fimmu.2020.00244. eCollection 2020. Front Immunol. 2020. PMID: 32153575 Free PMC article. Review.
References
-
- Armstrong DS, Grimwood K, Carlin JB, et al. Lower airway inflammation in infants and young children with cystic fibrosis. Am J Respir Crit Care Med. 1997;156:1197–204. - PubMed
-
- Hallett MB, Lloyds D. Neutrophil priming; the cellular signals which say ‘amber’ but not ‘green’. Immunol Today. 1995;16:264–8. - PubMed
-
- Khan TZ, Wagener JS, Bost T, et al. Early pulmonary inflammation in infants with cystic fibrosis. Am J Respir Crit Care Med. 1995;151:1075–82. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

