Mal3, the fission yeast EB1 homologue, cooperates with Bub1 spindle checkpoint to prevent monopolar attachment
- PMID: 16179942
- PMCID: PMC1369205
- DOI: 10.1038/sj.embor.7400540
Mal3, the fission yeast EB1 homologue, cooperates with Bub1 spindle checkpoint to prevent monopolar attachment
Abstract
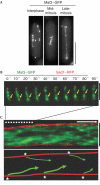
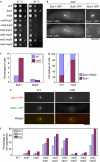
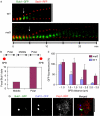
Bipolar microtubule attachment is central to genome stability. Here, we investigate the mitotic role of the fission yeast EB1 homologue Mal3. Mal3 shows dynamic inward movement along the spindle, initial emergence at the spindle pole body (SPB) and translocation towards the equatorial plane, followed by sudden disappearance. Deletion of Mal3 results in early mitotic delay, which is dependent on the Bub1, but not the Mad2, spindle checkpoint. Consistently, Bub1, but not Mad2, shows prolonged kinetochore localization. Double mutants between mal3 and a subset of checkpoint mutants, including bub1, bub3, mad3 and mph1, but not mad1 or mad2, show massive chromosome mis-segregation defects. In mal3bub1 mutants, both sister centromeres tend to remain in close proximity to one of the separating SPBs. Further analysis indicates that mis-segregated centromeres are exclusively associated with the mother SPB. Mal3, therefore, has a role in preventing monopolar attachment in cooperation with the Bub1/Bub3/Mad3/Mph1-dependent checkpoint.
Figures

References
-
- Browning H, Hackney DD, Nurse P (2003) Targeted movement of cell end factors in fission yeast. Nat Cell Biol 5: 812–818 - PubMed
-
- Busch KE, Brunner D (2004) The microtubule plus end-tracking proteins mal3p and tip1p cooperate for cell-end targeting of interphase microtubules. Curr Biol 14: 548–559 - PubMed
-
- Cleveland DW, Mao Y, Sullivan KF (2003) Centromeres and kinetochores: from epigenetics to mitotic checkpoint signaling. Cell 112: 407–421 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

