Cysteine protease mcII-Pa executes programmed cell death during plant embryogenesis
- PMID: 16183741
- PMCID: PMC1242326
- DOI: 10.1073/pnas.0506948102
Cysteine protease mcII-Pa executes programmed cell death during plant embryogenesis
Abstract
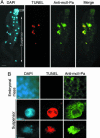
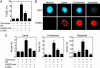
Programmed cell death (PCD) is indispensable for eukaryotic development. In animals, PCD is executed by the caspase family of cysteine proteases. Plants do not have close homologues of caspases but possess a phylogenetically distant family of cysteine proteases named metacaspases. The cellular function of metacaspases in PCD is unknown. Here we show that during plant embryogenesis, metacaspase mcII-Pa translocates from the cytoplasm to nuclei in terminally differentiated cells that are destined for elimination, where it colocalizes with the nuclear pore complex and chromatin, causing nuclear envelope disassembly and DNA fragmentation. The cell-death function of mcII-Pa relies on its cysteine-dependent arginine-specific proteolytic activity. Accordingly, mutation of catalytic cysteine abrogates the proteolytic activity of mcII-Pa and blocks nuclear degradation. These results establish metacaspase as an executioner of PCD during embryo patterning and provide a functional link between PCD and embryogenesis in plants. Although mcII-Pa and metazoan caspases have different substrate specificity, they serve a common function during development, demonstrating the evolutionary parallelism of PCD pathways in plants and animals.
Figures




References
-
- Baehrecke, E. (2002) Nat. Rev. Mol. Cell Biol. 3, 779-787. - PubMed
-
- Bozhkov, P. V., Suarez, M. F. & Filonova, L. H. (2005) Curr. Top. Dev. Biol. 67, 135-179. - PubMed
-
- Goldberg, R. B., de Paiva, G. & Yadegari, R. (1994) Science 266, 605-614. - PubMed
-
- Lukowitz, W., Roeder, A., Parmenter, D. & Somerville, C. (2004) Cell 116, 109-119. - PubMed
-
- Earnshaw, W. C. (1995) Curr. Opin. Cell Biol. 7, 337-343. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources

