Mating-induced shedding of cell walls, removal of walls from vegetative cells, and osmotic stress induce presumed cell wall genes in Chlamydomonas
- PMID: 16183845
- PMCID: PMC1256013
- DOI: 10.1104/pp.105.065037
Mating-induced shedding of cell walls, removal of walls from vegetative cells, and osmotic stress induce presumed cell wall genes in Chlamydomonas
Abstract
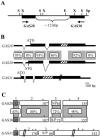
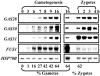
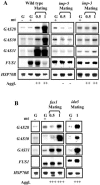
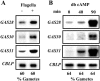
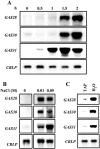
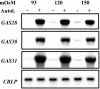
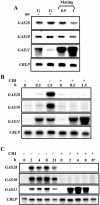
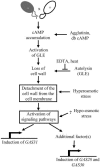
The first step in sexual differentiation of the unicellular green alga Chlamydomonas reinhardtii is the formation of gametes. Three genes, GAS28, GAS30, and GAS31, encoding Hyp-rich glycoproteins that presumably are cell wall constituents, are expressed in the late phase of gametogenesis. These genes, in addition, are activated by zygote formation and cell wall removal and by the application of osmotic stress. The induction by zygote formation could be traced to cell wall shedding prior to gamete fusion since it was seen in mutants defective in cell fusion. However, it was absent in mutants defective in the initial steps of mating, i.e. in flagellar agglutination and in accumulation of adenosine 3',5'-cyclic monophosphate in response to this agglutination. Induction of the three GAS genes was also observed when cultures were exposed to hypoosmotic or hyperosmotic stress. To address the question whether the induction seen upon cell wall removal from both gametes and vegetative cells was elicited by osmotic stress, cell wall removal was performed under isosmotic conditions. Also under such conditions an activation of the genes was observed, suggesting that the signaling pathway(s) is (are) activated by wall removal itself.
Figures




References
-
- Abe J, Kubo T, Takagi Y, Saito T, Miura K, Fukuzawa H, Matsuda Y (2004) The transcriptional program of synchronous gametogenesis in Chlamydomonas reinhardtii. Curr Genet 46: 304–315 - PubMed
-
- Adair WS, Hwang C, Goodenough UW (1983) Identification and visualization of the sexual agglutinin from the mating-type plus flagellar membrane of Chlamydomonas. Cell 33: 183–193 - PubMed
-
- Alonso-Monge R, Real E, Wojda I, Bebelman J-P, Mager WH, Siderius M (2001) Hyperosmotic stress response and regulation of cell wall integrity in Saccharomyces cerevisiae share common functional aspects. Mol Microbiol 4: 717–730 - PubMed
-
- Barlow JJ, Mathias AP, Williamson R (1963) A simple method for the quantitative isolation of undegraded high molecular weight ribonucleic acid. Biochem Biophys Res Commun 13: 61–66 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources

