Regulation of oxidative stress by the anti-aging hormone klotho
- PMID: 16186101
- PMCID: PMC2515369
- DOI: 10.1074/jbc.M509039200
Regulation of oxidative stress by the anti-aging hormone klotho
Abstract
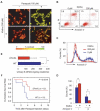
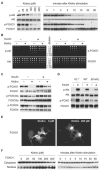
klotho is an aging suppressor gene and extends life span when overexpressed in mice. Klotho protein was recently demonstrated to function as a hormone that inhibits insulin/insulin-like growth factor-1 (IGF-1) signaling. Here we show that Klotho protein increases resistance to oxidative stress at the cellular and organismal level in mammals. Klotho protein activates the FoxO forkhead transcription factors that are negatively regulated by insulin/IGF-1 signaling, thereby inducing expression of manganese superoxide dismutase. This in turn facilitates removal of reactive oxygen species and confers oxidative stress resistance. Thus, Klotho-induced inhibition of insulin/IGF-1 signaling is associated with increased resistance to oxidative stress, which potentially contributes to the anti-aging properties of klotho.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

