TMC125 displays a high genetic barrier to the development of resistance: evidence from in vitro selection experiments
- PMID: 16188980
- PMCID: PMC1235844
- DOI: 10.1128/JVI.79.20.12773-12782.2005
TMC125 displays a high genetic barrier to the development of resistance: evidence from in vitro selection experiments
Abstract
TMC125 is a potent new investigational nonnucleoside reverse transcriptase inhibitor (NNRTI) that is active against human immunodeficiency virus type 1 (HIV-1) with resistance to currently licensed NNRTIs. Sequential passage experiments with both wild-type virus and NNRTI-resistant virus were performed to identify mutations selected by TMC125 in vitro. In addition to "classic" selection experiments at a low multiplicity of infection (MOI) with increasing concentrations of inhibitors, experiments at a high MOI with fixed concentrations of inhibitors were performed to ensure a standardized comparison between TMC125 and current NNRTIs. Both low- and high-MOI experiments demonstrated that the development of resistance to TMC125 required multiple mutations which frequently conferred cross-resistance to efavirenz and nevirapine. In high-MOI experiments, 1 muM TMC125 completely inhibited the breakthrough of resistant virus from wild-type and NNRTI-resistant HIV-1, in contrast to efavirenz and nevirapine. Furthermore, breakthrough of virus from site-directed mutant (SDM) SDM-K103N/Y181C occurred at the same time or later with TMC125 as breakthrough from wild-type HIV-1 with efavirenz or nevirapine. The selection experiments identified mutations selected by TMC125 that included known NNRTI-associated mutations L100I, Y181C, G190E, M230L, and Y318F and the novel mutations V179I and V179F. Testing the antiviral activity of TMC125 against a panel of SDMs indicated that the impact of these individual mutations on resistance was highly dependent upon the presence and identity of coexisting mutations. These results demonstrate that TMC125 has a unique profile of activity against NNRTI-resistant virus and possesses a high genetic barrier to the development of resistance in vitro.
Figures
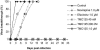

References
-
- Ait-Khaled, M., A. Rakik, P. Griffin, C. Stone, N. Richards, D. Thomas, J. Falloon, and M. Tisdale. 2003. HIV-1 reverse transcriptase and protease resistance mutations selected during 16-72 weeks of therapy in isolates from antiretroviral therapy-experienced patients receiving abacavir/efavirenz/amprenavir in the CNA2007 study. Antivir. Ther. 8:111-120. - PubMed
-
- Andries, K., H. Azijn, T. Thielemans, D. Ludovici, M. Kukla, J. Heeres, P. Janssen, B. De Corte, J. Vingerhoets, R. Pauwels, and M.-P. de Bethune. 2004. TMC125, a novel next-generation nonnucleoside reverse transcriptase inhibitor active against nonnucleoside reverse transcriptase inhibitor-resistant human immunodeficiency virus type 1. Antimicrob. Agents Chemother. 48:4680-4686. - PMC - PubMed
-
- Antinori, A., M. Zaccarelli, A. Cingolani, F. Forbici, M. G. Rizzo, M. P. Trotta, S. Di Giambenedetto, P. Narciso, A. Ammassari, E. Girardi, A. De Luca, and C. F. Perno. 2002. Cross-resistance among nonnucleoside reverse transcriptase inhibitors limits recycling efavirenz after nevirapine failure. AIDS Res. Hum. Retrovir. 18:835-838. - PubMed
-
- Bacheler, L., S. Jeffrey, G. Hanna, R. D'Aquila, L. Wallace, K. Logue, B. Cordova, K. Hertogs, B. Larder, R. Buckery, D. Baker, K. Gallagher, H. Scarnati, R. Tritch, and C. Rizzo. 2001. Genotypic correlates of phenotypic resistance to efavirenz in virus isolates from patients failing nonnucleoside reverse transcriptase inhibitor therapy. J. Virol. 75:4999-5008. - PMC - PubMed
-
- Bacheler, L. T., E. D. Anton, P. Kudish, D. Baker, J. Bunville, K. Krakowski, L. Bolling, M. Aujay, X. V. Wang, D. Ellis, M. F. Becker, A. L. Lasut, H. J. George, D. R. Spalding, G. Hollis, and K. Abremski. 2000. Human immunodeficiency virus type 1 mutations selected in patients failing efavirenz combination therapy. Antimicrob. Agents Chemother. 44:2475-2484. - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

