Oxadiazols: a new class of rationally designed anti-human immunodeficiency virus compounds targeting the nuclear localization signal of the viral matrix protein
- PMID: 16189005
- PMCID: PMC1235831
- DOI: 10.1128/JVI.79.20.13028-13036.2005
Oxadiazols: a new class of rationally designed anti-human immunodeficiency virus compounds targeting the nuclear localization signal of the viral matrix protein
Abstract
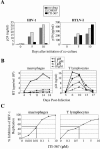
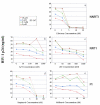
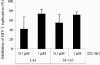
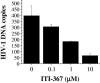
Despite recent progress in anti-human immunodeficiency virus (HIV) therapy, drug toxicity and emergence of drug-resistant isolates during long-term treatment of HIV-infected patients necessitate the search for new targets that can be used to develop novel antiviral agents. One such target is the process of nuclear translocation of the HIV preintegration complex. Previously we described a class of arylene bis(methylketone) compounds that inhibit HIV-1 nuclear import by targeting the nuclear localization signal (NLS) in the matrix protein (MA). Here we report a different class of MA NLS-targeting compounds that was selected using computer-assisted drug design. The leading compound from this group, ITI-367, showed potent anti-HIV activity in cultures of T lymphocytes and macrophages and also inhibited HIV-1 replication in ex vivo cultured lymphoid tissue. The virus carrying inactivating mutations in MA NLS was resistant to ITI-367. Analysis by real-time PCR demonstrated that the compound specifically inhibited nuclear import of viral DNA, measured by two-long terminal repeat circle formation. Evidence of the existence of this mechanism was provided by immunofluorescent microscopy, using fluorescently labeled HIV-1, which demonstrated retention of the viral DNA in the cytoplasm of drug-treated macrophages. Compounds inhibiting HIV-1 nuclear import may be attractive candidates for further development.
Figures

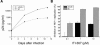

Similar articles
-
Backbone cyclic peptide, which mimics the nuclear localization signal of human immunodeficiency virus type 1 matrix protein, inhibits nuclear import and virus production in nondividing cells.Biochemistry. 1998 Apr 21;37(16):5616-22. doi: 10.1021/bi972878h. Biochemistry. 1998. PMID: 9548947
-
Nuclear localization signal of HIV-1 as a novel target for therapeutic intervention.Mol Med. 1995 Jan;1(2):217-30. Mol Med. 1995. PMID: 8529100 Free PMC article.
-
Two nuclear localization signals in the HIV-1 matrix protein regulate nuclear import of the HIV-1 pre-integration complex.J Mol Biol. 2000 Jun 2;299(2):359-68. doi: 10.1006/jmbi.2000.3768. J Mol Biol. 2000. PMID: 10860744
-
Blocking nuclear import of pre-integration complex: an emerging anti-HIV-1 drug discovery paradigm.Curr Med Chem. 2010;17(6):495-503. doi: 10.2174/092986710790416335. Curr Med Chem. 2010. PMID: 20015032 Review.
-
HIV-1 nuclear import: in search of a leader.Front Biosci. 1997 Dec 1;2:d578-87. doi: 10.2741/a213. Front Biosci. 1997. PMID: 9366553 Review.
Cited by
-
Discovery of a small-molecule antiviral targeting the HIV-1 matrix protein.Bioorg Med Chem Lett. 2013 Feb 15;23(4):1132-5. doi: 10.1016/j.bmcl.2012.11.041. Epub 2012 Nov 29. Bioorg Med Chem Lett. 2013. PMID: 23305922 Free PMC article.
-
Proteomic identification of differentially expressed and phosphorylated proteins in epidermis involved in larval-pupal metamorphosis of Helicoverpa armigera.BMC Genomics. 2009 Dec 12;10:600. doi: 10.1186/1471-2164-10-600. BMC Genomics. 2009. PMID: 20003373 Free PMC article.
-
Role of Autophagy in HIV-1 Matrix Protein p17-Driven Lymphangiogenesis.J Virol. 2017 Jul 27;91(16):e00801-17. doi: 10.1128/JVI.00801-17. Print 2017 Aug 15. J Virol. 2017. PMID: 28592537 Free PMC article.
-
HIV protein sequence hotspots for crosstalk with host hub proteins.PLoS One. 2011;6(8):e23293. doi: 10.1371/journal.pone.0023293. Epub 2011 Aug 15. PLoS One. 2011. PMID: 21858059 Free PMC article.
-
HIV-1 matrix protein p17 and its variants promote human triple negative breast cancer cell aggressiveness.Infect Agent Cancer. 2017 Sep 25;12:49. doi: 10.1186/s13027-017-0160-7. eCollection 2017. Infect Agent Cancer. 2017. PMID: 29021819 Free PMC article.
References
-
- Al Abed, Y., L. Dubrovsky, B. Ruzsicska, M. Seepersaud, and M. Bukrinsky. 2002. Inhibition of HIV-1 nuclear import via schiff base formation with arylene bis(methylketone) compounds. Bioorg. Med. Chem. Lett. 12:3117-3119. - PubMed
-
- Bouyac-Bertoia, M., J. D. Dvorin, R. A. Fouchier, Y. Jenkins, B. E. Meyer, L. I. Wu, M. Emerman, and M. H. Malim. 2001. Hiv-1 infection requires a functional integrase NLS. Mol. Cell 7:1025-1035. - PubMed
-
- Bowerman, B., P. O. Brown, J. M. Bishop, and H. E. Varmus. 1989. A nucleoprotein complex mediates the integration of retroviral DNA. Genes Dev. 3:469-478. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

