Canalicular immunostaining of aminopeptidase N (CD13) as a diagnostic marker for hepatocellular carcinoma
- PMID: 16189153
- PMCID: PMC1770740
- DOI: 10.1136/jcp.2005.026328
Canalicular immunostaining of aminopeptidase N (CD13) as a diagnostic marker for hepatocellular carcinoma
Abstract
Background: Aminopeptidase N (CD13) is expressed in normal and neoplastic liver tissue, where it exhibits a characteristic canalicular pattern (CD13(can)), similar to that seen for CD10 and when antibodies crossreact with biliary glycoprotein I (p-CEA).
Aim: To compare the putative diagnostic use of CD13(can) in differentiating between hepatocellular (HCC) and non-hepatocellular carcinomas metastatic to the liver (non-HCC).
Methods: A retrospective study comparing 53 HCC specimens with 32 non-HCC specimens. Immunostaining was performed with HepPar1 and antibodies directed against CD10, CD13, p-CEA, and alpha fetoprotein (AFP).
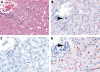
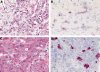
Results: In the HCC group, a canalicular staining pattern was found for CD13, p-CEA, and CD10 in 51, 43, and 33 specimens, respectively. HepPar1 was positive in 29 and AFP in 17 HCC specimens. In the non-HCC group, canalicular immunostaining for CD10 and p-CEA was confined to non-neoplastic liver tissue. One poorly differentiated cholangiocarcinoma showed apical expression of CD13, resembling to some extent CD13(can). Sensitivity and specificity were 96.2% and 97.0%, respectively, for CD13(can), 81.1% and 100% for p-CEA(can), 62.3% and 100%, for CD10(can), 54.7% and 99.9% for HepPar1, and 32.1% and 100% for AFP.
Conclusions: These results show that CD13(can) is more sensitive in differentiating between HCC and non-HCC than CD10(can), p-CEA(can), HepPar1, and AFP.
Figures


References
-
- D’Errico A, Baccarini P, Fiorentino M, et al. Histogenesis of primary liver carcinomas: strengths and weaknesses of cytokeratin profile and albumin mRNA detection. Hum Pathol 1996;27:599–604. - PubMed
-
- Hurlimann J, Gardiol D. Immunohistochemistry in the differential diagnosis of liver carcinomas. Am J Surg Pathol 1991;15:280–8. - PubMed
-
- Ma CK, Zarbo RJ, Frierson HF Jr, et al. Comparative immunohistochemical study of primary and metastatic carcinomas of the liver. Am J Clin Pathol 1993;99:551–7. - PubMed
-
- Koelma IA, Nap M, Huitema S, et al. Hepatocellular carcinoma, adenoma, and focal nodular hyperplasia. Comparative histopathologic study with immunohistochemical parameters. Arch Pathol Lab Med 1986;110:1035–40. - PubMed
-
- Wong MA, Yazdi HM. Hepatocellular carcinoma versus carcinoma metastatic to the liver. Value of stains for carcinoembryonic antigen and naphthylamidase in fine needle aspiration biopsy material. Acta Cytol 1990;34:192–6. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
