Genistein, a natural product from soy, is a potent inhibitor of transthyretin amyloidosis
- PMID: 16195386
- PMCID: PMC1253540
- DOI: 10.1073/pnas.0501609102
Genistein, a natural product from soy, is a potent inhibitor of transthyretin amyloidosis
Abstract
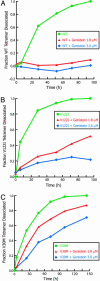
The misfolding of transthyretin (TTR), including rate-limiting tetramer dissociation and partial monomer denaturation, is sufficient for TTR misassembly into amyloid and other abnormal quaternary structures associated with three amyloid diseases: senile systemic amyloidosis, familial amyloid polyneuropathy, and familial amyloid cardiomyopathy. Small molecules can bind to one or both of the unoccupied TTR thyroid hormone-binding sites, stabilizing the native tetramer more than the dissociative transition state, thereby raising the kinetic barrier for tetramer dissociation. Herein we demonstrate that genistein, the major isoflavone natural product in soy, works in this fashion and is an excellent inhibitor of transthyretin tetramer dissociation and amyloidogenesis, reducing acid-mediated fibril formation to <10% of that exhibited by TTR alone. Genistein also inhibits the amyloidogenesis of the most common familial amyloid polyneuropathy and familial amyloid cardiomyopathy mutations in TTR: V30M and V122I, respectively. Genistein additionally inhibits tetramer dissociation under physiological conditions thought to lead to slow amyloidogenesis in humans. Furthermore, this natural product exhibits highly selective binding to TTR in plasma over all of the other plasma proteins. Isothermal titration calorimetry shows that genistein binds to TTR with negative cooperativity (K(d1) = 40 nM, K(d2) = 1.4 microM). The benefits of using a nutraceutical such as genistein to treat orphan diseases such as the TTR amyloidoses include known oral bioavailability and safety data. It is conceivable that some patients could benefit from simply increasing their intake of soy products or supplements.
Figures



References
-
- Setchell, K. D., Borriello, S. P., Hulme, P., Kirk, D. N. & Axelson, M. (1984) Am. J. Clin. Nutr. 40, 569-578. - PubMed
-
- Lee, H. P., Gourley, L., Duffy, S. W., Esteve, J., Lee, J. & Day, N. E. (1991) Lancet 337, 1197-1200. - PubMed
-
- Rose, D. P. (1992) Nutrition 8, 47-51. - PubMed
-
- Wu, A. H., Ziegler, R. G., Horn-Ross, P. L., Nomura, A. M., West, D. W., Kolonel, L. N., Rosenthal, J. F., Hoover, R. N. & Pike, M. C. (1996) Cancer Epidemiol. Biomarkers Prev. 5, 901-906. - PubMed
-
- Fukutake, M., Takahashi, M., Ishida, K., Kawamura, H., Sugimura, T. & Wakabayashi, K. (1996) Food Chem. Toxicol. 34, 457-461. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

