Identification of a novel extracellular cation-sensing G-protein-coupled receptor
- PMID: 16199532
- PMCID: PMC1435382
- DOI: 10.1074/jbc.M505186200
Identification of a novel extracellular cation-sensing G-protein-coupled receptor
Abstract
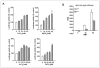
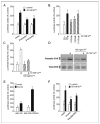
The C family G-protein-coupled receptors contain members that sense amino acid and extracellular cations, of which calcium-sensing receptor (CASR) is the prototypic extracellular calcium-sensing receptor. Some cells, such as osteoblasts in bone, retain responsiveness to extracellular calcium in CASR-deficient mice, consistent with the existence of another calcium-sensing receptor. We examined the calcium-sensing properties of GPRC6A, a newly identified member of this family. Alignment of GPRC6A with CASR revealed conservation of both calcium and calcimimetic binding sites. In addition, calcium, magnesium, strontium, aluminum, gadolinium, and the calcimimetic NPS 568 resulted in a dose-dependent stimulation of GPRC6A overexpressed in human embryonic kidney cells 293 cells. Also, osteocalcin, a calcium-binding protein highly expressed in bone, dose-dependently stimulated GPRC6A activity in the presence of calcium but inhibited the calcium-dependent activation of CASR. Coexpression of beta-arrestins 1 and 2, regulators of G-protein signaling RGS2 or RGS4, the RhoA inhibitor C3 toxin, the dominant negative Galpha(q)-(305-359) minigene, and pretreatment with pertussis toxin inhibited activation of GPRC6A by extracellular cations. Reverse transcription-PCR analyses showed that mouse GPRC6A is widely expressed in mouse tissues, including bone, calvaria, and the osteoblastic cell line MC3T3-E1. These data suggest that in addition to sensing amino acids, GPRC6A is a cation-, calcimimetic-, and osteocalcin-sensing receptor and a candidate for mediating extracellular calcium-sensing responses in osteoblasts and possibly other tissues.
Figures




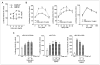
Similar articles
-
Osteoblast calcium-sensing receptor has characteristics of ANF/7TM receptors.J Cell Biochem. 2005 Aug 15;95(6):1081-92. doi: 10.1002/jcb.20500. J Cell Biochem. 2005. PMID: 15962313 Free PMC article.
-
A novel cation-sensing mechanism in osteoblasts is a molecular target for strontium.J Bone Miner Res. 2004 May;19(5):862-9. doi: 10.1359/JBMR.040114. Epub 2004 Jan 12. J Bone Miner Res. 2004. PMID: 15068510
-
Calcyclin mediates serum response element (SRE) activation by an osteoblastic extracellular cation-sensing mechanism.J Bone Miner Res. 2003 Oct;18(10):1825-33. doi: 10.1359/jbmr.2003.18.10.1825. J Bone Miner Res. 2003. PMID: 14584893
-
Multiligand specificity and wide tissue expression of GPRC6A reveals new endocrine networks.Endocrinology. 2012 May;153(5):2062-9. doi: 10.1210/en.2011-2117. Epub 2012 Feb 28. Endocrinology. 2012. PMID: 22374969 Free PMC article. Review.
-
Pharmacology and physiological function of the orphan GPRC6A receptor.Basic Clin Pharmacol Toxicol. 2020 Jun;126 Suppl 6:77-87. doi: 10.1111/bcpt.13397. Epub 2020 Mar 3. Basic Clin Pharmacol Toxicol. 2020. PMID: 32056382 Review.
Cited by
-
The calcium-sensing receptor in bone metabolism: from bench to bedside and back.Osteoporos Int. 2015 Aug;26(8):2055-71. doi: 10.1007/s00198-015-3203-1. Epub 2015 Jun 23. Osteoporos Int. 2015. PMID: 26100412 Review.
-
Receptors responsive to protein breakdown products in g-cells and d-cells of mouse, swine and human.Front Physiol. 2012 Apr 10;3:65. doi: 10.3389/fphys.2012.00065. eCollection 2012. Front Physiol. 2012. PMID: 22514536 Free PMC article.
-
Osteocalcin: A Multifaceted Bone-Derived Hormone.Annu Rev Nutr. 2023 Aug 21;43:55-71. doi: 10.1146/annurev-nutr-061121-091348. Annu Rev Nutr. 2023. PMID: 37603430 Free PMC article. Review.
-
The GPRC6A receptor displays constitutive internalization and sorting to the slow recycling pathway.J Biol Chem. 2017 Apr 28;292(17):6910-6926. doi: 10.1074/jbc.M116.762385. Epub 2017 Mar 9. J Biol Chem. 2017. PMID: 28280242 Free PMC article.
-
Genomic and non-genomic effects of androgens in the cardiovascular system: clinical implications.Clin Sci (Lond). 2017 Jul 1;131(13):1405-1418. doi: 10.1042/CS20170090. Clin Sci (Lond). 2017. PMID: 28645930 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

