Scopolamine reduces persistent activity related to long-term encoding in the parahippocampal gyrus during delayed matching in humans
- PMID: 16207870
- PMCID: PMC6725748
- DOI: 10.1523/JNEUROSCI.1982-05.2005
Scopolamine reduces persistent activity related to long-term encoding in the parahippocampal gyrus during delayed matching in humans
Abstract
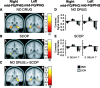
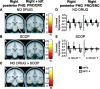
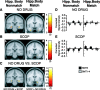
Recent computational modeling and slice physiology studies have suggested that long-term encoding may depend on sustained spiking during brief memory delays in parahippocampal neurons, and that this persistent spiking activity is modulated by effects of acetylcholine at muscarinic receptors. Our recent functional magnetic resonance imaging (fMRI) study has shown that sustained parahippocampal delay period activity during delayed match-to-sample performance in healthy young individuals predicted subsequent memory of visual stimuli on a recognition memory assessment 20 min later (Schon et al., 2004). The current study combined this fMRI paradigm with a pharmacological manipulation to test whether this long-term encoding-related delay activity is reduced in subjects who receive the muscarinic cholinergic antagonist scopolamine before fMRI scanning. Subsequent memory was predicted by sustained activity during brief memory delays bilaterally in the perirhinal/entorhinal cortex, in the right posterior parahippocampal and mid-fusiform gyri, and in the hippocampal body in healthy young individuals without a scopolamine challenge. This activity was reduced in subjects receiving scopolamine. The results are consistent with computational modeling data and behavioral pharmacological studies, suggesting that long-term encoding-related activity may be reduced if cholinergic receptors are blocked by scopolamine.
Figures

Similar articles
-
Paradoxical facilitation of object recognition memory after infusion of scopolamine into perirhinal cortex: implications for cholinergic system function.J Neurosci. 2006 Sep 13;26(37):9520-9. doi: 10.1523/JNEUROSCI.2319-06.2006. J Neurosci. 2006. PMID: 16971536 Free PMC article.
-
Scopolamine disrupts hippocampal activity during allocentric spatial memory in humans: an fMRI study using a virtual reality analogue of the Morris Water Maze.J Psychopharmacol. 2011 Sep;25(9):1256-65. doi: 10.1177/0269881110379285. Epub 2010 Sep 7. J Psychopharmacol. 2011. PMID: 20823079 Clinical Trial.
-
Persistence of parahippocampal representation in the absence of stimulus input enhances long-term encoding: a functional magnetic resonance imaging study of subsequent memory after a delayed match-to-sample task.J Neurosci. 2004 Dec 8;24(49):11088-97. doi: 10.1523/JNEUROSCI.3807-04.2004. J Neurosci. 2004. PMID: 15590925 Free PMC article.
-
The interactive effect of acute ovarian suppression and the cholinergic system on visuospatial working memory in young women.Psychoneuroendocrinology. 2010 Aug;35(7):987-1000. doi: 10.1016/j.psyneuen.2009.12.012. Epub 2010 Jan 25. Psychoneuroendocrinology. 2010. PMID: 20102786 Clinical Trial.
-
Mechanisms underlying working memory for novel information.Trends Cogn Sci. 2006 Nov;10(11):487-93. doi: 10.1016/j.tics.2006.09.005. Epub 2006 Oct 2. Trends Cogn Sci. 2006. PMID: 17015030 Free PMC article. Review.
Cited by
-
Reduced spiking in entorhinal cortex during the delay period of a cued spatial response task.Learn Mem. 2012 May 15;19(6):219-30. doi: 10.1101/lm.025866.112. Learn Mem. 2012. PMID: 22589278 Free PMC article.
-
The cholinergic hypothesis of cognitive aging revisited again: cholinergic functional compensation.Pharmacol Biochem Behav. 2011 Aug;99(2):254-61. doi: 10.1016/j.pbb.2011.02.022. Epub 2011 Mar 5. Pharmacol Biochem Behav. 2011. PMID: 21382398 Free PMC article. Review.
-
The prominent role of stimulus processing: cholinergic function and dysfunction in cognition.Curr Opin Neurol. 2011 Aug;24(4):364-70. doi: 10.1097/WCO.0b013e328348bda5. Curr Opin Neurol. 2011. PMID: 21725241 Free PMC article.
-
Advances in functional magnetic resonance imaging: technology and clinical applications.Neurotherapeutics. 2007 Jul;4(3):360-70. doi: 10.1016/j.nurt.2007.05.007. Neurotherapeutics. 2007. PMID: 17599702 Free PMC article. Review.
-
Flexible modulation of network connectivity related to cognition in Alzheimer's disease.Neuroimage. 2014 Oct 15;100:544-57. doi: 10.1016/j.neuroimage.2014.05.032. Epub 2014 May 20. Neuroimage. 2014. PMID: 24852459 Free PMC article.
References
-
- Aigner TG, Mishkin M (1986) The effects of physostigmine and scopolamine on recognition memory in monkeys. Behav Neural Biol 45: 81-87. - PubMed
-
- Aigner TG, Walker DL, Mishkin M (1991) Comparison of the effects of scopolamine administered before and after acquisition in a test of visual recognition memory in monkeys. Behav Neural Biol 55: 61-67. - PubMed
-
- Atri A, Sherman S, Norman KA, Kirchhoff BA, Nicolas MM, Greicius MD, Cramer SC, Breiter HC, Hasselmo ME, Stern CE (2004) Blockade of central cholinergic receptors impairs new learning and increases proactive interference in a word paired-associate memory task. Behav Neurosci 118: 223-236. - PubMed
-
- Awh E, Jonides J (2001) Overlapping mechanisms of attention and spatial working memory. Trends Cogn Sci 5: 119-126. - PubMed
-
- Bartus RT, Johnson HR (1976) Short-term memory in the rhesus monkey: disruption from the anti-cholinergic scopolamine. Pharmacol Biochem Behav 5: 39-46. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
