Effect of incorporating a 10 minute point of care test for salivary nicotine metabolites into a general practice based smoking cessation programme: randomised controlled trial
- PMID: 16210250
- PMCID: PMC1273454
- DOI: 10.1136/bmj.38621.463900.7C
Effect of incorporating a 10 minute point of care test for salivary nicotine metabolites into a general practice based smoking cessation programme: randomised controlled trial
Abstract
Objective: To investigate the effect of immediate feedback from a point of care test for salivary nicotine metabolites in promoting smoking cessation and reduction in tobacco use.
Design: Prospective, operator blinded, randomised controlled trial.
Setting: General dental practice, London.
Participants: 100 adult smokers.
Interventions: Participants completed a questionnaire on smoking, undertook a clinical examination, and received counselling in smoking cessation. Saliva samples were analysed at presentation and at eight weeks for salivary nicotine metabolites using a 10 minute semiquantitative point of care test.
Main outcome measures: Smoking cessation measured by salivary nicotine metabolite values (scale 0-6), patient feedback on the perceived value of the test (visual analogue scale) in quitting, and reduction in tobacco use.
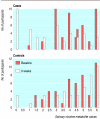
Results: A higher smoking quit rate was achieved with the point of care test (23% cases v 7% controls; P < 0.039), and overall tobacco use also decreased (68% cases v 28% controls; P < 0.001). Baseline values for salivary nicotine metabolites did not differ between the groups (cases, mean 4.1, SD 1.3 and 4.3, 1.4; P = 0.51). 87 participants reattended at eight weeks (44 cases, 43 controls). Mean nicotine metabolite values at eight weeks were 2.58 (2.0) for cases and 4.29 (1.8) for controls (P < 0.001).
Conclusion: Incorporation of individualised personal feedback using a point of care test for salivary nicotine metabolites into a general practice based smoking cessation programme increased quit rates by 17% at eight weeks and reduced tobacco use.
Figures
Comment in
-
Near patient tests for smoking cessation.BMJ. 2005 Oct 29;331(7523):979-80. doi: 10.1136/bmj.331.7523.979. BMJ. 2005. PMID: 16254278 Free PMC article. No abstract available.
References
-
- World Health Organization. Annual report from WHO's tobacco free initiative. Geneva: WHO, 1999.
-
- Tobacco Advisory Group of the Royal College of Physicians. Nicotine addiction in Britain [updated 2001]. www.rcplondon.ac.uk (accessed Jan 2003).
-
- Peto R, Lopez AD, Boreham J. Updated national and international estimates of tobacco-attributed mortality. www.ctsu.ox.ac.uk (accessed Mar 2004).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical