p600, a unique protein required for membrane morphogenesis and cell survival
- PMID: 16214886
- PMCID: PMC1247991
- DOI: 10.1073/pnas.0507458102
p600, a unique protein required for membrane morphogenesis and cell survival
Erratum in
- Proc Natl Acad Sci U S A. 2005 Dec 6;102(49):17882
Abstract
In this article, we identify and characterize p600, a unique 600-kDa retinoblastoma protein- and calmodulin-binding protein. In the nucleus, p600 and retinoblastoma protein seem to act as a chromatin scaffold. In the cytoplasm, p600 and clathrin form a meshwork structure, which could contribute to cytoskeletal organization and membrane morphogenesis. Reduced expression of p600 with interference RNA abrogates integrin-mediated ruffled membrane formation and, furthermore, prevents activation of integrin-mediated survival pathways. Consequently, knockdown of p600 sensitizes cells to apoptosis induced by cell detachment. These findings provide mechanistic insight into the regulation of membrane-proximal events in tumorigenesis.
Figures

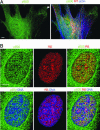

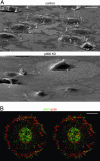

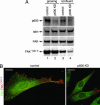
References
-
- Hahn, W. C. & Weinberg, R. A. (2002) Nat. Rev. Cancer 2, 331–341. - PubMed
-
- Vogelstein, B. & Kinzler, K. W. (2004) Nat. Med. 10, 789–799. - PubMed
-
- Nevins, J. R. (2001) in Fields Virology, eds. Knipe, D. M. & Howley, P. M. (Lippincott Williams & Wilkins, Philadelphia), 4th Ed., pp. 245–283.
-
- Muller, H. & Helin, K. (2000) Biochim. Biophys. Acta 1470, M1–M12. - PubMed
-
- Jones, D. L., Thompson, D. A. & Munger, K. (1997) Virology 239, 97–107. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

