Signal propagation from membrane messengers to nuclear effectors revealed by reporters of phosphoinositide dynamics and Akt activity
- PMID: 16214892
- PMCID: PMC1257695
- DOI: 10.1073/pnas.0502889102
Signal propagation from membrane messengers to nuclear effectors revealed by reporters of phosphoinositide dynamics and Akt activity
Abstract
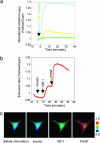
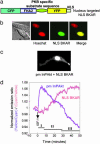
Among various second messengers, phosphatidylinositol 3,4,5-triphosphate (PIP3) and phosphatidylinositol 3,4-bisphosphate [PI(3,4)P2] regulate a variety of cellular processes, such as cell survival, polarization, and proliferation. Many of these functions are achieved via activation of serine/threonine kinase Akt. To investigate the spatiotemporal regulation of these lipids, we constructed a genetically targetable phosphoinositide (PI) indicator by sandwiching pleckstrin homology (PH) domain of Akt and a "pseudoligand" containing acidic amino acid residues, between cyan and yellow mutants of GFP. In living cells, elevations in PIP3 and PI(3,4)P2 by growth factor-induced activation of phosphatidylinositol 3-kinase (PI3K) resulted in a change in fluorescence resonance energy transfer (FRET) between the fluorescent proteins, increasing yellow to cyan emission ratios by 10-30%. This response can be reversed by inhibiting PI3K and abolished by mutating the critical residues responsible for PI binding. Differential dynamics of PIs were observed at plasma membrane of NIH 3T3 cells, stimulated by various growth factors. On the other hand, the nuclear targeted indicator showed no response within an hour after platelet-derived growth factor stimulation, suggesting that no appreciable amounts of accessible PIP3 and PI(3,4)P2 were produced in the nucleus. Furthermore, simultaneous imaging of a plasma membrane-targeted PI indicator and a nuclear-targeted Akt activity reporter revealed a gradual and sustained accumulation of Akt activity in the nucleus after rapid and transient production of PIP3 and PI(3,4)P2 at plasma membrane in the same cell. Thus, signal propagation from the lipid messengers at plasma membrane to the effectors in the nucleus is precisely controlled by kinases as well as lipid and protein phosphatases.
Figures


References
-
- Martin, T. F. (1998) Annu. Rev. Cell Dev. Biol. 14, 231–264. - PubMed
-
- Fruman, D. A., Meyers, R. E. & Cantley, L. C. (1998) Annu. Rev. Biochem. 67, 481–507. - PubMed
-
- Maehama, T., Taylor, G. S. & Dixon, J. E. (2001) Annu. Rev. Biochem. 70, 247–279. - PubMed
-
- Krystal, G. (2000) Semin. Immunol. 12, 397–403. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

