A phase II trial of vaccination with autologous, tumor-derived heat-shock protein peptide complexes Gp96, in combination with GM-CSF and interferon-alpha in metastatic melanoma patients
- PMID: 16215718
- PMCID: PMC11031093
- DOI: 10.1007/s00262-005-0084-8
A phase II trial of vaccination with autologous, tumor-derived heat-shock protein peptide complexes Gp96, in combination with GM-CSF and interferon-alpha in metastatic melanoma patients
Abstract
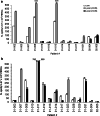
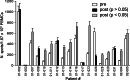
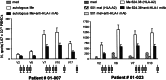
The aim of this study was to determine the immunogenicity and antitumor activity of autologous, tumor-derived heat shock protein gp96-peptide complex vaccine (HSPPC-96; Oncophage given with GM-CSF and IFN-alpha in pre-treated metastatic (AJCC stage IV) melanoma patients. Patients underwent surgical resection of metastatic lesions for HSPPC-96 production. HSPPC-96 was administered subcutaneously (s.c.) in four weekly intervals (first cycle). Patients with more available vaccine and absence of progressive disease received four additional injections in 2-week intervals (second cycle) or more. GM-CSF was given s.c. at the same site at days -1, 0 and +1, while IFN-alpha (3 MU) was administered s.c. at a different site at days +4 and +6. Antigen-specific anti-melanoma T and NK lymphocyte response was assessed by enzyme-linked immunospot assay on peripheral blood mononuclear cells obtained before and after vaccination. Thirty-eight patients were enrolled, 20 received at least four injections (one cycle) of HSPPC-96 and were considered assessable. Toxicity was mild and most treatment-related adverse events were local erythema and induration at the injection site. Patients receiving at least four injections of HSPPC-96 were considered evaluable for clinical response: of the 18 patients with measurable disease post surgery, 11 showed stable disease (SD). The ELISPOT assay revealed an increased class I HLA-restricted T and NK cell-mediated post-vaccination response in 5 out of 17 and 12 out of the 18 patients tested, respectively. Four of the five class I HLA-restricted T cell responses fall in the group of SD patients. Vaccination with autologous HSPPC-96 together with GM-CSF and IFN-alpha is feasible and accompanied by mild local and systemic toxicity. Both tumor-specific T cell-mediated and NK cell responses were generated in a proportion of patients. Clinical activity was limited to SD. However, both immunological and clinical responses were not improved as compared with those recorded in a previous study investigating HSPPC-96 monotherapy.
Figures
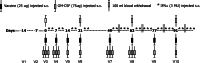

Similar articles
-
Vaccination of metastatic melanoma patients with autologous tumor-derived heat shock protein gp96-peptide complexes: clinical and immunologic findings.J Clin Oncol. 2002 Oct 15;20(20):4169-80. doi: 10.1200/JCO.2002.09.134. J Clin Oncol. 2002. PMID: 12377960 Clinical Trial.
-
Identification of a new subset of myeloid suppressor cells in peripheral blood of melanoma patients with modulation by a granulocyte-macrophage colony-stimulation factor-based antitumor vaccine.J Clin Oncol. 2007 Jun 20;25(18):2546-53. doi: 10.1200/JCO.2006.08.5829. J Clin Oncol. 2007. PMID: 17577033
-
Autologous tumor-derived heat-shock protein peptide complex-96 (HSPPC-96) in patients with metastatic melanoma.J Transl Med. 2010 Jan 29;8:9. doi: 10.1186/1479-5876-8-9. J Transl Med. 2010. PMID: 20109236 Free PMC article. Clinical Trial.
-
HSPPC-96 vaccine in metastatic melanoma patients: from the state of the art to a possible future.Expert Rev Vaccines. 2009 Nov;8(11):1513-26. doi: 10.1586/erv.09.108. Expert Rev Vaccines. 2009. PMID: 19863242 Review.
-
Immune modulations during chemoimmunotherapy & novel vaccine strategies--in metastatic melanoma and non small-cell lung cancer.Dan Med J. 2013 Dec;60(12):B4774. Dan Med J. 2013. PMID: 24355457 Review.
Cited by
-
Heat shock proteins expressed in the marsupial Tasmanian devil are potential antigenic candidates in a vaccine against devil facial tumour disease.PLoS One. 2018 Apr 27;13(4):e0196469. doi: 10.1371/journal.pone.0196469. eCollection 2018. PLoS One. 2018. PMID: 29702669 Free PMC article.
-
Myeloma cell line-derived, pooled heat shock proteins as a universal vaccine for immunotherapy of multiple myeloma.Blood. 2009 Oct 29;114(18):3880-9. doi: 10.1182/blood-2009-06-227355. Epub 2009 Aug 4. Blood. 2009. PMID: 19654406 Free PMC article.
-
Dendritic-tumor fusion cells derived heat shock protein70-peptide complex has enhanced immunogenicity.PLoS One. 2015 May 11;10(5):e0126075. doi: 10.1371/journal.pone.0126075. eCollection 2015. PLoS One. 2015. PMID: 25961716 Free PMC article.
-
Induction of leukemia-specific antibodies by immunotherapy with leukemia-cell-derived heat shock protein 70.Cancer Sci. 2008 Jul;99(7):1427-34. doi: 10.1111/j.1349-7006.2008.00829.x. Epub 2008 Apr 29. Cancer Sci. 2008. PMID: 18452562 Free PMC article.
-
High molecular weight stress proteins: Identification, cloning and utilisation in cancer immunotherapy.Int J Hyperthermia. 2013 Aug;29(5):364-75. doi: 10.3109/02656736.2013.803607. Epub 2013 Jul 5. Int J Hyperthermia. 2013. PMID: 23829534 Free PMC article. Review.
References
-
- Belli F, Testori A, Rivoltini L, Maio M, Andreola G, Sertoli MR, Gallino P, Piris A, Cattelan A, Lazzari I, Carrabba M, Scita G, et al. Vaccination of metastatic melanoma patients with autologous tumor-derived heat shock protein gp96-peptide complexes: Clinical and immunologic findings. J Clin Oncol. 2002;20:4169–4180. doi: 10.1200/JCO.2002.09.134. - DOI - PubMed
-
- Inaba K, Inaba M, Romani N, Aya H, Deguchi M, Ikehara S, Muramatsu S, Steinman RM. Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte-macrophage colony stimulating factor. J Exp Med. 1992;176:1693–1699. doi: 10.1084/jem.176.6.1693. - DOI - PMC - PubMed
-
- Soiffer R, Lynch T, Mihm M, Jung K, Rhuda C, Schmollinger JC, Hodi FS, Liebster L, Lam P, Mentzer S, Singer S, Tanabe KK, et al. Vaccination with irradiated autologous melanoma cells engineered to secrete human granulocyte-macrophage-colony stimulating factor generates potent antitumor immunity in patients with metastatic melanoma. Proc Natl Acad Sci USA. 1998;95:13141–13146. doi: 10.1073/pnas.95.22.13141. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

