Protein phosphatase 2A is a negative regulator of IL-2 production in patients with systemic lupus erythematosus
- PMID: 16224536
- PMCID: PMC1253625
- DOI: 10.1172/JCI24895
Protein phosphatase 2A is a negative regulator of IL-2 production in patients with systemic lupus erythematosus
Abstract
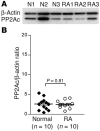
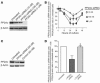
Decreased IL-2 production in systemic lupus erythematosus (SLE) represents a central component of the disease immunopathology. We report that the message, protein, and enzymatic activity of the catalytic subunit of protein phosphatase 2A (PP2Ac), but not PP1, are increased in patients with SLE regardless of disease activity and treatment and in a disease-specific manner. Treatment of SLE T cells with PP2Ac-siRNA decreased the protein levels and activity of PP2Ac in a specific manner and increased the levels of phosphorylated cAMP response element-binding protein and its binding to the IL2 and c-fos promoters, as well as increased activator protein 1 activity, causing normalization of IL-2 production. Our data document increased activity of PP2A as a novel SLE disease-specific abnormality and define a distinct mechanism whereby it represses IL-2 production. We propose the use of PP2Ac-siRNA as a novel tool to correct T cell IL-2 production in SLE patients.
Figures



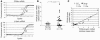

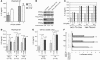

Similar articles
-
Systemic lupus erythematosus serum IgG increases CREM binding to the IL-2 promoter and suppresses IL-2 production through CaMKIV.J Clin Invest. 2005 Apr;115(4):996-1005. doi: 10.1172/JCI22854. J Clin Invest. 2005. PMID: 15841182 Free PMC article.
-
Antisense cyclic adenosine 5'-monophosphate response element modulator up-regulates IL-2 in T cells from patients with systemic lupus erythematosus.J Immunol. 2002 Oct 15;169(8):4147-52. doi: 10.4049/jimmunol.169.8.4147. J Immunol. 2002. PMID: 12370343
-
[Interleukin-2 signaling pathway regulating molecules in systemic lupus erythematosus].Beijing Da Xue Xue Bao Yi Xue Ban. 2016 Dec 18;48(6):1100-1104. Beijing Da Xue Xue Bao Yi Xue Ban. 2016. PMID: 27987522 Review. Chinese.
-
Correcting the expression of miRNA-155 represses PP2Ac and enhances the release of IL-2 in PBMCs of juvenile SLE patients.Lupus. 2015 Mar;24(3):240-7. doi: 10.1177/0961203314552117. Epub 2014 Sep 24. Lupus. 2015. PMID: 25253569
-
The serine/threonine protein phosphatase 2A controls autoimmunity.Clin Immunol. 2018 Jan;186:38-42. doi: 10.1016/j.clim.2017.07.012. Epub 2017 Jul 21. Clin Immunol. 2018. PMID: 28736280 Free PMC article. Review.
Cited by
-
Induction of PP2A Bβ, a regulator of IL-2 deprivation-induced T-cell apoptosis, is deficient in systemic lupus erythematosus.Proc Natl Acad Sci U S A. 2011 Jul 26;108(30):12443-8. doi: 10.1073/pnas.1103915108. Epub 2011 Jul 11. Proc Natl Acad Sci U S A. 2011. PMID: 21746932 Free PMC article.
-
SLE-Associated Defects Promote Altered T Cell Function.Crit Rev Immunol. 2017;37(1):39-58. doi: 10.1615/CritRevImmunol.2018025213. Crit Rev Immunol. 2017. PMID: 29431078 Free PMC article. Review.
-
PP2A dephosphorylates Elf-1 and determines the expression of CD3zeta and FcRgamma in human systemic lupus erythematosus T cells.J Immunol. 2008 Sep 1;181(5):3658-64. doi: 10.4049/jimmunol.181.5.3658. J Immunol. 2008. PMID: 18714041 Free PMC article.
-
Mechanisms for suppression of interleukin-6 expression in peritoneal macrophages from docosahexaenoic acid-fed mice.J Nutr Biochem. 2009 May;20(5):358-68. doi: 10.1016/j.jnutbio.2008.04.006. Epub 2008 Jul 7. J Nutr Biochem. 2009. PMID: 18602807 Free PMC article.
-
Epigenetic mechanisms in lupus.Curr Opin Rheumatol. 2010 Sep;22(5):478-82. doi: 10.1097/BOR.0b013e32833ae915. Curr Opin Rheumatol. 2010. PMID: 20445453 Free PMC article. Review.
References
-
- Linker-Israeli M, et al. Defective production of interleukin 1 and interleukin 2 in patients with systemic lupus erythematosus (SLE) J. Immunol. 1983;130:2651–2655. - PubMed
-
- Theofilopoulos AN, Dixon FJ. Murine models of systemic lupus erythematosus. Adv. Immunol. 1985;37:269–390. - PubMed
-
- Nelson BH. IL-2, regulatory T cells, and tolerance. J. Immunol. 2004;172:3983–3988. - PubMed
-
- Iliopoulos AG, Tsokos GC. Immunopathogenesis and spectrum of infections in systemic lupus erythematosus. Semin. Arthritis Rheum. 1996;25:318–336. - PubMed
-
- Kovacs B, Vassilopoulos D, Vogelgesang SA, Tsokos GC. Defective CD3-mediated cell death in activated T cells from patients with systemic lupus erythematosus: role of decreased intracellular TNF-alpha. Clin. Immunol. Immunopathol. 1996;81:293–302. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

