Induction of macrophage-derived SLPI by Mycobacterium tuberculosis depends on TLR2 but not MyD88
- PMID: 16236128
- PMCID: PMC1802419
- DOI: 10.1111/j.1365-2567.2005.02238.x
Induction of macrophage-derived SLPI by Mycobacterium tuberculosis depends on TLR2 but not MyD88
Abstract
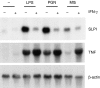
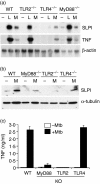
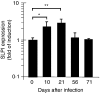
Macrophages respond to Mycobacterium tuberculosis by regulating expression of gene products that initiate a host innate response to this micro-organism. In this study, we report that exposure of murine peritoneal macrophages to heat-killed Mycobacterium tuberculosis (HK-Mtb) led to an increase in secretory leucocyte protease inhibitor (SLPI) gene expression and protein secretion in a time- and dose-dependent manner. HK-Mtb-induced SLPI mRNA expression was sensitive neither to a protein synthesis inhibitor, cycloheximide, nor to an actin polymerization blocker, cytochalasin D. Treatment of macrophages with interferon (IFN)-gamma inhibited HK-Mtb-induced SLPI expression. RAW264.7 cells stably expressing SLPI produced a reduced level of tumour necrosis factor (TNF) in response to HK-Mtb as compared with mock transfectants. Aerosol infection of mice with live M. tuberculosis resulted in an induction of SLPI gene expression in infected lungs. Macrophages from Toll-like receptor 4 (TLR4)-/- or MyD88-/- mice responded to M. tuberculosis similarly to wild-type macrophages by exhibiting increased SLPI expression. In contrast, macrophages from TLR2-/- mice were incapable of inducing SLPI in response to M. tuberculosis. This induction signifies the presence of a TLR2-dependent but MyD88-independent M. tuberculosis signalling pathway, suggesting involvement of adaptor protein(s) other than MyD88 in M. tuberculosis-mediated induction of SLPI.
Figures


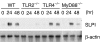

Similar articles
-
Expression of many immunologically important genes in Mycobacterium tuberculosis-infected macrophages is independent of both TLR2 and TLR4 but dependent on IFN-alphabeta receptor and STAT1.J Immunol. 2005 Sep 1;175(5):3318-28. doi: 10.4049/jimmunol.175.5.3318. J Immunol. 2005. PMID: 16116224
-
MyD88 and TLR2, but not TLR4, are required for host defense against Cryptococcus neoformans.Eur J Immunol. 2005 Mar;35(3):870-8. doi: 10.1002/eji.200425799. Eur J Immunol. 2005. PMID: 15714580
-
Toll-like receptor 2 mediates inflammatory cytokine induction but not sensitization for liver injury by Propioni- bacterium acnes.J Leukoc Biol. 2005 Dec;78(6):1255-64. doi: 10.1189/jlb.0804448. Epub 2005 Oct 4. J Leukoc Biol. 2005. PMID: 16204620
-
Toll-like receptor 2 in host defense against Mycobacterium tuberculosis: to be or not to be-that is the question.Curr Opin Immunol. 2016 Oct;42:76-82. doi: 10.1016/j.coi.2016.06.003. Epub 2016 Jun 18. Curr Opin Immunol. 2016. PMID: 27326654 Free PMC article. Review.
-
Toll-Like Receptor 4 as an Immune Receptor Against Mycobacterium tuberculosis: A Systematic Review.Lab Med. 2019 Apr 8;50(2):117-129. doi: 10.1093/labmed/lmy047. Lab Med. 2019. PMID: 30124945
Cited by
-
Divergent Functions of TLR2 on Hematopoietic and Nonhematopoietic Cells during Chronic Mycobacterium tuberculosis Infection.J Immunol. 2017 Jan 15;198(2):741-748. doi: 10.4049/jimmunol.1601651. Epub 2016 Dec 5. J Immunol. 2017. PMID: 27920273 Free PMC article.
-
Herpes simplex virus downregulates secretory leukocyte protease inhibitor: a novel immune evasion mechanism.J Virol. 2008 Oct;82(19):9337-44. doi: 10.1128/JVI.00603-08. Epub 2008 Jul 30. J Virol. 2008. PMID: 18667508 Free PMC article.
-
The impact of IFN-γ receptor on SLPI expression in active tuberculosis: association with disease severity.Am J Pathol. 2014 May;184(5):1268-73. doi: 10.1016/j.ajpath.2014.01.006. Epub 2014 Mar 4. Am J Pathol. 2014. PMID: 24606882 Free PMC article.
-
TRAM is required for TLR2 endosomal signaling to type I IFN induction.J Immunol. 2014 Dec 15;193(12):6090-102. doi: 10.4049/jimmunol.1401605. Epub 2014 Nov 10. J Immunol. 2014. PMID: 25385819 Free PMC article.
-
Role of interferon-γ and inflammatory monocytes in driving colonic inflammation during acute Clostridium difficile infection in mice.Immunology. 2017 Apr;150(4):468-477. doi: 10.1111/imm.12700. Epub 2017 Jan 30. Immunology. 2017. PMID: 27995603 Free PMC article.
References
-
- Barton GM, Medzhitov R. Toll-like receptor signaling pathways. Science. 2003;300:1524–5. - PubMed
-
- O'Neill LA, Fitzgerald KA, Bowie AG. The Toll-IL-1 receptor adaptor family grows to five members. Trends Immunol. 2003;24:286–90. - PubMed
-
- Modlin RL. Activation of toll-like receptors by microbial lipoproteins: role in host defense. J Allergy Clin Immunol. 2001;108:S104–6. - PubMed
-
- Horng T, Barton GM, Medzhitov R. TIRAP: an adapter molecule in the Toll signaling pathway. Nat Immunol. 2001;2:835–41. - PubMed
-
- Yamamoto M, Sato S, Hemmi H, et al. Essential role for TIRAP in activation of the signalling cascade shared by TLR2 and TLR4. Nature. 2002;420:324–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

