A mycothiol synthase mutant of Mycobacterium smegmatis produces novel thiols and has an altered thiol redox status
- PMID: 16237013
- PMCID: PMC1272995
- DOI: 10.1128/JB.187.21.7309-7316.2005
A mycothiol synthase mutant of Mycobacterium smegmatis produces novel thiols and has an altered thiol redox status
Abstract
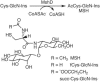
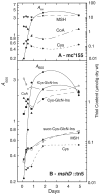
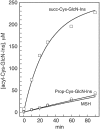
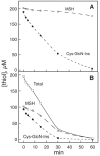
Mycobacteria and other actinomycetes do not produce glutathione but make mycothiol (MSH; AcCys-GlcN-Ins) that has functions similar to those of glutathione and is essential for growth of Mycobacterium tuberculosis. Mycothiol synthase (MshD) catalyzes N acetylation of Cys-GlcN-Ins to produce MSH in Mycobacterium smegmatis mc2155, and Cys-GlcN-Ins is maintained at a low level. The mycothiol synthase mutant, the mshD::Tn5 mutant, produces high levels of Cys-GlcN-Ins along with two novel thiols, N-formyl-Cys-GlcN-Ins and N-succinyl-Cys-GlcN-Ins, and a small amount of MSH. The nonenzymatic reaction of acyl-coenzyme A (CoA) with Cys-GlcN-Ins to produce acyl-Cys-GlcN-Ins is a facile reaction under physiologic conditions, with succinyl-CoA being an order of magnitude more reactive than acetyl-CoA. The uncatalyzed reaction rates are adequate to account for the observed production of N-succinyl-Cys-GlcN-Ins and MSH under physiologic conditions. It was shown that the N-acyl-Cys-GlcN-Ins compounds are maintained in a substantially reduced state in the mutant but that Cys-GlcN-Ins exists in disulfide forms at 5 to 40% at different stages of growth. MSH was able to facilitate reduction of N-succinyl-Cys-GlcN-Ins disulfide through thiol-disulfide exchange, but N-formyl-Cys-GlcN-Ins was ineffective. The oxidized state of Cys-GlcN-Ins in cells appears to result from a high susceptibility to autoxidation and a low capacity of the cell to reduce its disulfide forms. The mutant exhibited no enhanced sensitivity to hydrogen peroxide, tert-butyl hydroperoxide, or cumene hydroperoxide relative to the parent strain, suggesting that the most abundant thiol, N-formyl-Cys-GlcN-Ins, functions as a substitute for MSH.
Figures

Similar articles
-
Identification of the mycothiol synthase gene (mshD) encoding the acetyltransferase producing mycothiol in actinomycetes.Arch Microbiol. 2002 Nov;178(5):331-7. doi: 10.1007/s00203-002-0462-y. Epub 2002 Aug 15. Arch Microbiol. 2002. PMID: 12375100
-
Mycothiol biosynthesis and metabolism. Cellular levels of potential intermediates in the biosynthesis and degradation of mycothiol in mycobacterium smegmatis.J Biol Chem. 1998 Nov 13;273(46):30391-7. doi: 10.1074/jbc.273.46.30391. J Biol Chem. 1998. PMID: 9804803
-
A mycothiol synthase mutant of Mycobacterium tuberculosis has an altered thiol-disulfide content and limited tolerance to stress.J Bacteriol. 2006 Sep;188(17):6245-52. doi: 10.1128/JB.00393-06. J Bacteriol. 2006. PMID: 16923891 Free PMC article.
-
Mycothiol biochemistry.Arch Microbiol. 2002 Dec;178(6):388-94. doi: 10.1007/s00203-002-0469-4. Epub 2002 Sep 3. Arch Microbiol. 2002. PMID: 12420157 Review.
-
Chemistry and Redox Biology of Mycothiol.Antioxid Redox Signal. 2018 Feb 20;28(6):487-504. doi: 10.1089/ars.2017.7074. Epub 2017 May 10. Antioxid Redox Signal. 2018. PMID: 28372502 Review.
Cited by
-
Structures and mechanisms of the mycothiol biosynthetic enzymes.Curr Opin Chem Biol. 2009 Oct;13(4):451-9. doi: 10.1016/j.cbpa.2009.07.018. Epub 2009 Aug 19. Curr Opin Chem Biol. 2009. PMID: 19699138 Free PMC article. Review.
-
Mycothiol import by Mycobacterium smegmatis and function as a resource for metabolic precursors and energy production.J Bacteriol. 2007 Oct;189(19):6796-805. doi: 10.1128/JB.00644-07. Epub 2007 Jul 20. J Bacteriol. 2007. PMID: 17644601 Free PMC article.
-
Organic hydroperoxide resistance protein and ergothioneine compensate for loss of mycothiol in Mycobacterium smegmatis mutants.J Bacteriol. 2011 Apr;193(8):1981-90. doi: 10.1128/JB.01402-10. Epub 2011 Feb 18. J Bacteriol. 2011. PMID: 21335456 Free PMC article.
-
Drug targets in mycobacterial sulfur metabolism.Infect Disord Drug Targets. 2007 Jun;7(2):140-58. doi: 10.2174/187152607781001772. Infect Disord Drug Targets. 2007. PMID: 17970225 Free PMC article. Review.
-
Identification of Resistance Genes and Response to Arsenic in Rhodococcus aetherivorans BCP1.Front Microbiol. 2019 May 7;10:888. doi: 10.3389/fmicb.2019.00888. eCollection 2019. Front Microbiol. 2019. PMID: 31133997 Free PMC article.
References
-
- Beckwith, J., and F. Aslund. 2001. Roles of thiol-redox pathways in bacteria. Annu. Rev. Biochem. 55:21-48. - PubMed
-
- Berges, D. A., W. E. DeWolf, Jr., G. L. Dunn, D. J. Newman, S. J. Schmidt, J. J. Taggart, and C. Gilvarg. 1986. Studies on the active site of succinyl-CoA:tetrahydrodipicolinate N-succinyltransferase. Characterization using analogs of tetrahydrodipicolinate. J. Biol. Chem. 261:6160-6167. - PubMed
-
- Buchmeier, N. A., G. L. Newton, T. Koledin, and R. C. Fahey. 2003. Association of mycothiol with protection of Mycobacterium tuberculosis from toxic oxidants and antibiotics. Mol. Microbiol. 47:1723-1732. - PubMed
-
- Deutsch, J., and H. J. Niclas. 1993. A convenient procedure for the formylation of amines and alcohols using cyanomethylformate. Synthetic Commun. 23:1561-1568.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

