Identification of a new neuropeptide precursor reveals a novel source of extrinsic modulation in the feeding system of Aplysia
- PMID: 16237168
- PMCID: PMC6725720
- DOI: 10.1523/JNEUROSCI.2932-05.2005
Identification of a new neuropeptide precursor reveals a novel source of extrinsic modulation in the feeding system of Aplysia
Abstract
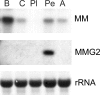
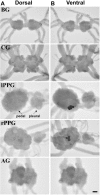
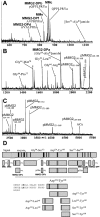
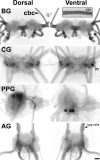
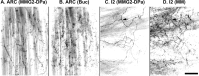
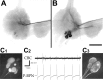
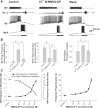
The Aplysia feeding system is advantageous for investigating the role of neuropeptides in behavioral plasticity. One family of Aplysia neuropeptides is the myomodulins (MMs), originally purified from one of the feeding muscles, the accessory radula closer (ARC). However, two MMs, MMc and MMe, are not encoded on the only known MM gene. Here, we identify MM gene 2 (MMG2), which encodes MMc and MMe and four new neuropeptides. We use matrix-assisted laser desorption/ionization time-of-flight mass spectrometry to verify that these novel MMG2-derived peptides (MMG2-DPs), as well as MMc and MMe, are synthesized from the precursor. Using antibodies against the MMG2-DPs, we demonstrate that neuronal processes that stain for MMG2-DPs are found in the buccal ganglion, which contains the feeding network, and in the buccal musculature including the ARC muscle. Surprisingly, however, no immunostaining is observed in buccal neurons including the ARC motoneurons. In situ hybridization reveals only few MMG2-expressing neurons that are mostly located in the pedal ganglion. Using immunohistochemical and electrophysiological techniques, we demonstrate that some of these pedal neurons project to the buccal ganglion and are the likely source of the MMG2-DP innervation of the feeding network and musculature. We show that the MMG2-DPs are bioactive both centrally and peripherally: they bias egestive feeding programs toward ingestive ones, and they modulate ARC muscle contractions. The multiple actions of the MMG2-DPs suggest that these peptides play a broad role in behavioral plasticity and that the pedal-buccal projection neurons that express them are a novel source of extrinsic modulation of the feeding system of Aplysia.
Figures

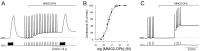

Similar articles
-
Discovery of leucokinin-like neuropeptides that modulate a specific parameter of feeding motor programs in the molluscan model, Aplysia.J Biol Chem. 2017 Nov 17;292(46):18775-18789. doi: 10.1074/jbc.M117.795450. Epub 2017 Sep 18. J Biol Chem. 2017. PMID: 28924050 Free PMC article.
-
Identification and characterization of the feeding circuit-activating peptides, a novel neuropeptide family of aplysia.J Neurosci. 2002 Sep 1;22(17):7797-808. doi: 10.1523/JNEUROSCI.22-17-07797.2002. J Neurosci. 2002. PMID: 12196603 Free PMC article.
-
The enterins: a novel family of neuropeptides isolated from the enteric nervous system and CNS of Aplysia.J Neurosci. 2001 Oct 15;21(20):8247-61. doi: 10.1523/JNEUROSCI.21-20-08247.2001. J Neurosci. 2001. PMID: 11588196 Free PMC article.
-
Feeding neural networks in the mollusc Aplysia.Neurosignals. 2004 Jan-Apr;13(1-2):70-86. doi: 10.1159/000076159. Neurosignals. 2004. PMID: 15004426 Review.
-
Physiology and biochemistry of peptidergic cotransmission in Aplysia.J Physiol Paris. 1993;87(3):141-51. doi: 10.1016/0928-4257(93)90025-o. J Physiol Paris. 1993. PMID: 7907908 Review.
Cited by
-
Identification and functional characterization of the first molluscan neuromedin U receptor in the slug, Deroceras reticulatum.Sci Rep. 2020 Dec 18;10(1):22308. doi: 10.1038/s41598-020-79047-x. Sci Rep. 2020. PMID: 33339848 Free PMC article.
-
Mass spectrometry imaging and profiling of single cells.J Proteomics. 2012 Aug 30;75(16):5036-5051. doi: 10.1016/j.jprot.2012.03.017. Epub 2012 Mar 29. J Proteomics. 2012. PMID: 22498881 Free PMC article. Review.
-
Neuromodulation of neuronal circuits: back to the future.Neuron. 2012 Oct 4;76(1):1-11. doi: 10.1016/j.neuron.2012.09.010. Neuron. 2012. PMID: 23040802 Free PMC article. Review.
-
Luqin-like RYamide peptides regulate food-evoked responses in C. elegans.Elife. 2017 Aug 29;6:e28877. doi: 10.7554/eLife.28877. Elife. 2017. PMID: 28847365 Free PMC article.
-
Urotensin II in invertebrates: from structure to function in Aplysia californica.PLoS One. 2012;7(11):e48764. doi: 10.1371/journal.pone.0048764. Epub 2012 Nov 8. PLoS One. 2012. PMID: 23144960 Free PMC article.
References
-
- Brezden BL, Yeoman MS, Gardner DR, Benjamin PR (1999) FMRFamideactivated Ca2+ channels in Lymnaea heart cells are modulated by “SEEPLY,” a neuropeptide encoded on the same gene. J Neurophysiol 81: 1818-1826. - PubMed
-
- Brezina V, Bank B, Cropper EC, Rosen S, Vilim FS, Kupfermann I, Weiss KR (1995) Nine members of the myomodulin family of peptide cotransmitters at the B16-ARC neuromuscular junction of Aplysia J Neurophysiol 74: 54-72. - PubMed
-
- Brezina V, Orekhova IV, Weiss KR (1996) Functional uncoupling of linked neurotransmitter effects by combinatorial convergence. Science 273: 806-810. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
