Engagement of CD83 ligand induces prolonged expansion of CD8+ T cells and preferential enrichment for antigen specificity
- PMID: 16239433
- PMCID: PMC1895397
- DOI: 10.1182/blood-2005-05-2073
Engagement of CD83 ligand induces prolonged expansion of CD8+ T cells and preferential enrichment for antigen specificity
Abstract
Following T-cell receptor and CD28 signaling, CD8+ T cells express a receptor for CD83, a molecule up-regulated on functionally mature dendritic cells. Although this expression pattern suggests that CD83 is involved in adaptive immunity, little is known about its function in the periphery, and the existence of its ligand on T cells is controversial. We demonstrate that the engagement of the CD83 ligand (CD83L) preferentially enriches and significantly amplifies the number of antigen-specific CD8+ T cells. Coengagement of the T-cell receptor, CD28, and CD83L supports priming of naive CD8+ T cells that retain antigen specificity and cytotoxic function for more than 6 months. Therefore, engagement of the CD83L provides a unique signal to activated CD8+ T cells that could be exploited to generate long-lived antigen-specific cytotoxic T cells for the treatment of cancer and infection.
Figures
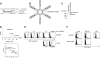

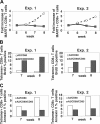
 indicates A2; □, A2/CD80;
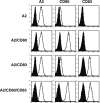
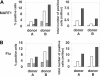
indicates A2; □, A2/CD80;  , A2/CD83; ▪, A2/CD80/CD83. (B) Likewise, when APCs are pulsed with Flu peptide, the percentage and total number of antigen-specific T cells was increased when T cells were stimulated by APC/A2/CD80 (percentage, P = .009; number, P = .01) and APC/A2/CD80/CD83 (percentage, P = .009; number, P = .023), but not by or APC/A2/CD83 (percentage, P = .32; number, P = .32). When compared with APC/A2/CD80, APC/A2/CD80/CD83 generates a significant increase in the percentage and number of Flu-specific T cells (percentage, P = .01; number, P = .035).
, A2/CD83; ▪, A2/CD80/CD83. (B) Likewise, when APCs are pulsed with Flu peptide, the percentage and total number of antigen-specific T cells was increased when T cells were stimulated by APC/A2/CD80 (percentage, P = .009; number, P = .01) and APC/A2/CD80/CD83 (percentage, P = .009; number, P = .023), but not by or APC/A2/CD83 (percentage, P = .32; number, P = .32). When compared with APC/A2/CD80, APC/A2/CD80/CD83 generates a significant increase in the percentage and number of Flu-specific T cells (percentage, P = .01; number, P = .035).  indicates A2; □, A2/CD80;
indicates A2; □, A2/CD80;  , A2/CD83; ▪, A2/CD80/CD83.
, A2/CD83; ▪, A2/CD80/CD83.
Similar articles
-
Urban particulate matter stimulation of human dendritic cells enhances priming of naive CD8 T lymphocytes.Immunology. 2018 Apr;153(4):502-512. doi: 10.1111/imm.12852. Epub 2017 Nov 28. Immunology. 2018. PMID: 29044495 Free PMC article.
-
Activated T cells induce rapid CD83 expression on B cells by engagement of CD40.Immunol Lett. 2011 May;136(2):221-7. doi: 10.1016/j.imlet.2011.01.013. Epub 2011 Jan 28. Immunol Lett. 2011. PMID: 21277328
-
Dendritic cell CD83: a therapeutic target or innocent bystander?Immunol Lett. 2008 Jan 15;115(1):1-8. doi: 10.1016/j.imlet.2007.10.001. Epub 2007 Oct 29. Immunol Lett. 2008. PMID: 18001846 Free PMC article. Review.
-
Endogenous Expression of the Human CD83 Attenuates EAE Symptoms in Humanized Transgenic Mice and Increases the Activity of Regulatory T Cells.Front Immunol. 2019 Jun 25;10:1442. doi: 10.3389/fimmu.2019.01442. eCollection 2019. Front Immunol. 2019. PMID: 31293592 Free PMC article.
-
CD83 regulates lymphocyte maturation, activation and homeostasis.Trends Immunol. 2008 Apr;29(4):186-94. doi: 10.1016/j.it.2008.01.009. Epub 2008 Mar 7. Trends Immunol. 2008. PMID: 18329338 Review.
Cited by
-
A peptide antigen derived from EGFR T790M is immunogenic in non‑small cell lung cancer.Int J Oncol. 2015 Feb;46(2):497-504. doi: 10.3892/ijo.2014.2787. Epub 2014 Dec 1. Int J Oncol. 2015. PMID: 25532027 Free PMC article.
-
Artificial antigen-presenting cells: the booster for the obtaining of functional adoptive cells.Cell Mol Life Sci. 2024 Aug 31;81(1):378. doi: 10.1007/s00018-024-05412-y. Cell Mol Life Sci. 2024. PMID: 39215816 Free PMC article. Review.
-
CD83 expression characterizes precursor exhausted T cell population.Commun Biol. 2023 Mar 11;6(1):258. doi: 10.1038/s42003-023-04631-6. Commun Biol. 2023. PMID: 36906640 Free PMC article.
-
Impaired tumor antigen processing by immunoproteasome-expressing CD40-activated B cells and dendritic cells.Cancer Immunol Immunother. 2011 Jun;60(6):857-67. doi: 10.1007/s00262-011-0995-5. Epub 2011 Mar 13. Cancer Immunol Immunother. 2011. PMID: 21400024 Free PMC article.
-
RGMb is a novel binding partner for PD-L2 and its engagement with PD-L2 promotes respiratory tolerance.J Exp Med. 2014 May 5;211(5):943-59. doi: 10.1084/jem.20130790. Epub 2014 Apr 21. J Exp Med. 2014. PMID: 24752301 Free PMC article.
References
-
- van Lier RA, ten Berge IJ, Gamadia LE. Human CD8(+) T-cell differentiation in response to viruses. Nat Rev Immunol. 2003;3: 931-939. - PubMed
-
- Steinman RM. Some interfaces of dendritic cell biology. Apmis. 2003;111: 675-697. - PubMed
-
- O'Neill DW, Adams S, Bhardwaj N. Manipulating dendritic cell biology for the active immunotherapy of cancer. Blood. 2004;104: 2235-2246. - PubMed
-
- Acuto O, Michel F. CD28-mediated co-stimulation: a quantitative support for TCR signalling. Nat Rev Immunol. 2003;3: 939-951. - PubMed
-
- Zhou LJ, Schwarting R, Smith HM, Tedder TF. A novel cell-surface molecule expressed by human interdigitating reticulum cells, Langerhans cells, and activated lymphocytes is a new member of the Ig superfamily. J Immunol. 1992;149: 735-742. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

