Immune responses against a single CD8+-T-cell epitope induced by virus vector vaccination can successfully control Trypanosoma cruzi infection
- PMID: 16239534
- PMCID: PMC1273883
- DOI: 10.1128/IAI.73.11.7356-7365.2005
Immune responses against a single CD8+-T-cell epitope induced by virus vector vaccination can successfully control Trypanosoma cruzi infection
Abstract
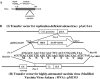
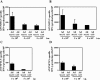
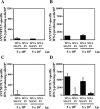
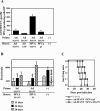
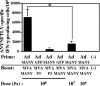
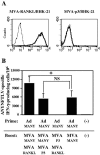
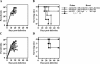
In order to develop CD8+-T-cell-mediated immunotherapy against intracellular infectious agents, vaccination using recombinant virus vectors has become a promising strategy. In this study, we generated recombinant adenoviral and vaccinia virus vectors expressing a single CD8+-T-cell epitope, ANYNFTLV, which is derived from a Trypanosoma cruzi antigen. Immunogenicity of these two recombinant virus vectors was confirmed by the detection of ANYNFTLV-specific CD8+ T cells in the spleens of immunized mice. Priming/boosting immunization using combinations of these two recombinant virus vectors revealed that the adenovirus vector was efficient for priming and the vaccinia virus vector was effective for boosting the CD8+-T-cell responses. Moreover, we also demonstrated that the ANYNFTLV-specific CD8+-T-cell responses were further augmented by coadministration of recombinant vaccinia virus vector expressing the receptor activator of NFkappaB (RANK) ligand as an adjuvant. By priming with the adenovirus vector expressing ANYNFTLV and boosting with the vaccinia virus vectors expressing ANYNFTLV and RANK ligand, the immunized mice were efficiently protected from subsequent challenge with lethal doses of T. cruzi. These results indicated, for the first time, that the induction of immune responses against a single CD8+-T-cell epitope derived from an intrinsic T. cruzi antigen was sufficient to control lethal T. cruzi infection.
Figures
References
-
- Anderson, D. M., E. Maraskovsky, W. L. Billingsley, W. C. Dougall, M. E. Tometsko, E. R. Roux, M. C. Teepe, R. F. DuBose, D. Cosman, and L. Galibert. 1997. A homologue of the TNF receptor and its ligand enhance T-cell growth and dendritic-cell function. Nature 390:175-179. - PubMed
-
- Brener, Z., and R. T. Gazzinelli. 1997. Immunological control of Trypanosoma cruzi infection and pathogenesis of Chagas' disease. Int. Arch. Allergy Immunol. 114:103-110. - PubMed
-
- Bruna-Romero, O., G. Gonzalez-Aseguinolaza, J. C. Hafalla, M. Tsuji, and R. S. Nussenzweig. 2001. Complete, long-lasting protection against malaria of mice primed and boosted with two distinct virus vectors expressing the same plasmodial antigen. Proc. Natl. Acad. Sci. USA 98:11491-11496. - PMC - PubMed
-
- Buus, S., and M. H. Claesson. 2004. Identifying multiple tumor-specific epitopes from large-scale screening for overexpressed mRNA. Curr. Opin. Immunol. 16:137-142. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

