Infiltration of COX-2-expressing macrophages is a prerequisite for IL-1 beta-induced neovascularization and tumor growth
- PMID: 16239969
- PMCID: PMC1257532
- DOI: 10.1172/JCI23298
Infiltration of COX-2-expressing macrophages is a prerequisite for IL-1 beta-induced neovascularization and tumor growth
Abstract
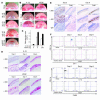
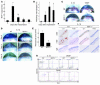
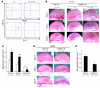
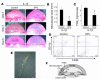
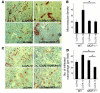
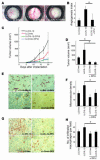
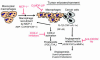
Inflammatory angiogenesis is a critical process in tumor progression and other diseases. The inflammatory cytokine IL-1beta promotes angiogenesis, tumor growth, and metastasis, but its mechanisms remain unclear. We examined the association between IL-1beta-induced angiogenesis and cell inflammation. IL-1beta induced neovascularization in the mouse cornea at rates comparable to those of VEGF. Neutrophil infiltration occurred on day 2. Macrophage infiltration occurred on days 4 and 6. The anti-Gr-1 Ab-induced depletion of infiltrating neutrophils did not affect IL-1beta- or VEGF-induced angiogenesis. The former was reduced in monocyte chemoattractant protein-1-deficient (MCP-1(-/-)) mice compared with wild-type mice. After day 4, clodronate liposomes, which kill macrophages, reduced IL-1beta-induced angiogenesis and partially inhibited VEGF-induced angiogenesis. Infiltrating macrophages near the IL-1beta-induced neovasculature were COX-2 positive. Lewis lung carcinoma cells expressing IL-1beta (LLC/IL-1beta) developed neovasculature with macrophage infiltration and enhanced tumor growth in wild-type but not MCP-1(-/-) mice. A COX-2 inhibitor reduced tumor growth, angiogenesis, and macrophage infiltration in LLC/IL-1beta. Thus, macrophage involvement might be a prerequisite for IL-1beta-induced neovascularization and tumor progression.
Figures


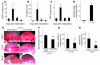
References
-
- Carmeliet P. Angiogenesis in health and disease. Nat. Med. 2003;9:653–660. - PubMed
-
- Rafii S, Lyden D. Therapeutic stem and progenitor cell transplantation for organ vascularization and regeneration. Nat. Med. 2003;9:702–712. - PubMed
-
- Dinarello CA. Biologic basis for interleukin-1 in disease. Blood. 1996;87:2095–2147. - PubMed
-
- Mantovani A, Bussolino F, Dejana E. Cytokine regulation of endothelial cell function. FASEB J. 1992;6:2591–2599. - PubMed
-
- Torisu H, et al. Macrophage infiltration correlates with tumor stage and angiogenesis in human malignant melanoma: possible involvement of TNFalpha and IL-1alpha. Int. J. Cancer. 2000;85:182–188. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

