Life in the balance: how BH3-only proteins induce apoptosis
- PMID: 16243507
- PMCID: PMC2930980
- DOI: 10.1016/j.ceb.2005.10.001
Life in the balance: how BH3-only proteins induce apoptosis
Abstract
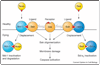
BH3-only members of the Bcl-2 intracellular protein family, which include Bim, Bmf, Bik, Bad, Bid, Puma, Noxa and Hrk, mediate many developmentally programmed and induced cytotoxic signals. They have key roles in development, tissue homeostasis, immunity and tumor suppression, and compounds mimicking them are promising anti-cancer agents. Their activity is normally constrained by transcriptional and/or diverse post-transcriptional controls. When activated, these death ligands engage pro-survival Bcl-2-like proteins via the BH3 domain, inactivating their function. Bim and Puma bind all the pro-survival proteins, whereas others, such as Noxa and Bad, engage distinct subsets and exhibit complementary killing. Hence, multiple pro-survival proteins must be inactivated to unleash Bax and Bak, which drive apoptosis. Whether certain BH3-only proteins also directly activate Bax/Bak remains controversial.
Figures


References
-
-
Cory S, Huang DCS, Adams JM. The Bcl-2 family: roles in cell survival and oncogenesis. Oncogene. 2003;22:8590–8607. A useful review on the Bcl-2 family and its role in cancer.
-
-
-
Danial NN, Korsmeyer SJ. Cell death: critical control points. Cell. 2004;116:205–219. A good recent overview of apoptosis.
-
-
- Cheng EH, Wei MC, Weiler S, Flavell RA, Mak TW, Lindsten T, Korsmeyer SJ. BCL-2, BCL-xL sequester BH3 domain-only molecules preventing BAX- and BAK-mediated mitochondrial apoptosis. Mol Cell. 2001;8:705–711. - PubMed
-
- Green DR, Kroemer G. The pathophysiology of mitochondrial cell death. Science. 2004;305:626–629. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

