Anti-human immunodeficiency virus type 1 activity of the nonnucleoside reverse transcriptase inhibitor GW678248 in combination with other antiretrovirals against clinical isolate viruses and in vitro selection for resistance
- PMID: 16251284
- PMCID: PMC1280120
- DOI: 10.1128/AAC.49.11.4465-4473.2005
Anti-human immunodeficiency virus type 1 activity of the nonnucleoside reverse transcriptase inhibitor GW678248 in combination with other antiretrovirals against clinical isolate viruses and in vitro selection for resistance
Abstract
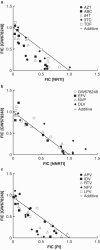
GW678248, a novel nonnucleoside reverse transcriptase inhibitor, has been evaluated for anti-human immunodeficiency virus activity in a variety of in vitro assays against laboratory strains and clinical isolates. When GW678248 was tested in combination with approved drugs in the nucleoside and nucleotide reverse transcriptase inhibitor classes or the protease inhibitor class, the antiviral activities were either synergistic or additive. When GW678248 was tested in combination with approved drugs in the nonnucleoside reverse transcriptase inhibitor class, the antiviral activities were either additive or slightly antagonistic. Clinical isolates from antiretroviral drug-experienced patients were selected for evaluation of sensitivity to GW678248 in a recombinant virus assay. Efavirenz (EFV) and nevirapine (NVP) had > or = 10-fold increases in their 50% inhibitory concentrations (IC50s) for 85% and 98% of the 55 selected isolates, respectively, whereas GW678248 had a > or = 10-fold increase in the IC50 for only 17% of these isolates. Thus, 81 to 83% of the EFV- and/or NVP-resistant viruses from this data set were susceptible to GW678248. Virus populations resistant to GW678248 were selected by in vitro dose-escalating serial passage. Resistant progeny viruses recovered after eight passages had amino acid substitutions V106I, E138K, and P236L in the reverse transcriptase-coding region in one passage series and amino acid substitutions K102E, V106A, and P236L in a second passage series.
Figures
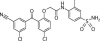

Similar articles
-
Antiviral activity of GW678248, a novel benzophenone nonnucleoside reverse transcriptase inhibitor.Antimicrob Agents Chemother. 2005 Oct;49(10):4046-51. doi: 10.1128/AAC.49.10.4046-4051.2005. Antimicrob Agents Chemother. 2005. PMID: 16189079 Free PMC article.
-
In vitro selection of mutations in human immunodeficiency virus type 1 reverse transcriptase that confer resistance to capravirine, a novel nonnucleoside reverse transcriptase inhibitor.Antiviral Res. 2006 Jun;70(2):66-74. doi: 10.1016/j.antiviral.2006.01.001. Epub 2006 Jan 25. Antiviral Res. 2006. PMID: 16472877
-
Structure-activity relationship studies of novel benzophenones leading to the discovery of a potent, next generation HIV nonnucleoside reverse transcriptase inhibitor.J Med Chem. 2006 Jan 26;49(2):727-39. doi: 10.1021/jm050670l. J Med Chem. 2006. PMID: 16420058
-
Efavirenz a nonnucleoside reverse transcriptase inhibitor of first-generation: Approaches based on its medicinal chemistry.Eur J Med Chem. 2016 Jan 27;108:455-465. doi: 10.1016/j.ejmech.2015.11.025. Epub 2015 Nov 25. Eur J Med Chem. 2016. PMID: 26708112 Review.
-
The emerging profile of cross-resistance among the nonnucleoside HIV-1 reverse transcriptase inhibitors.Viruses. 2014 Jul 31;6(8):2960-73. doi: 10.3390/v6082960. Viruses. 2014. PMID: 25089538 Free PMC article. Review.
Cited by
-
[Evaluation of antiretroviral therapy on mother to child transmission HIV in HIV-1 positive pregnant women: case of St. Camillus Medical Center in Ouagadougou, Burkina Faso].Pan Afr Med J. 2015 Apr 23;20:399. doi: 10.11604/pamj.2015.20.399.5627. eCollection 2015. Pan Afr Med J. 2015. PMID: 26301003 Free PMC article. French.
-
Antiviral activity of GW678248, a novel benzophenone nonnucleoside reverse transcriptase inhibitor.Antimicrob Agents Chemother. 2005 Oct;49(10):4046-51. doi: 10.1128/AAC.49.10.4046-4051.2005. Antimicrob Agents Chemother. 2005. PMID: 16189079 Free PMC article.
-
Status Presens of Antiviral Drugs And Strategies: Part I: DNA Viruses and Retroviruses.Adv Antivir Drug Des. 2007;5:1-58. doi: 10.1016/S1075-8593(06)05001-5. Epub 2007 Sep 2. Adv Antivir Drug Des. 2007. PMID: 32288472 Free PMC article. Review.
-
Potent inhibition of HIV replication in primary human cells by novel synthetic polyketides inspired by Aureothin.Sci Rep. 2020 Jan 28;10(1):1326. doi: 10.1038/s41598-020-57843-9. Sci Rep. 2020. PMID: 31992748 Free PMC article.
-
Cell-permeable stapled peptides based on HIV-1 integrase inhibitors derived from HIV-1 gene products.ACS Chem Biol. 2013 Oct 18;8(10):2235-44. doi: 10.1021/cb400495h. Epub 2013 Aug 15. ACS Chem Biol. 2013. PMID: 23898787 Free PMC article.
References
-
- The AIDS Clinical Trials Group Virology Technical Advisory Committee and the Division of AIDS, National Institutes of Allergy and Infectious Diseases. 1994. HIV drug susceptibility assay. ACTG virology manual for HIV laboratories. RES 4-5. Division of AIDS, National Institutes of Allergy and Infectious Diseases, Bethesda, Md.
-
- Andries, K., H. Azijn, T. Thielemans, D. Ludovici, M. Kukla, J. Heeres, P. Janssen, B. De Corte, J. Vingerhoets, R. Pauwels, and M. de Bethune. 2004. TMC125, a novel next-generation nonnucleoside reverse transcriptase inhibitor active against nonnucleoside reverse transcriptase inhibitor-resistant human immunodeficiency virus type 1. Antimicrob. Agents Chemother. 48:4680-4686. - PMC - PubMed
-
- Bacheler, L. T., E. D. Anton, P. Kudish, D. Baker, J. Burnville, K. Krakowski, L. Bolling, M. Aujay, X. V. Wang, D. Ellis, M. F. Becker, A. L. Lasut, H. J. George, D. R. Spalding, G. Hollis, and K. Abremski. 2000. Human immunodeficiency virus type 1 mutation selected in patients failing efavirenz combination therapy. Antimicrob. Agents Chemother. 44:2475-2484. - PMC - PubMed
-
- Chan, J. H., G. A. Freeman, J. H. Tidwell, K. R. Romines, L. T. Schaller, J. R. Cowan, S. S. Gonzales, G. S. Lowell, C. W. Andrews, D. J. Reynolds, M. St. Clair, R. J. Hazen, R. G. Ferris, K. L. Creech, G. B. Roberts, S. A. Short, K. Weaver, G. W. Koszalka, and L. R. Boone. 2004. Novel benzophenones as non-nucleoside reverse transcriptase inhibitors of HIV-1. J. Med. Chem. 47:1175-1182. - PubMed
-
- Daluge, S. M., D. J. M. Purifoy, P. M. Savina, M. H. St. Clair, N. R. Parry, I. K. Dev, P. Novak, K. M. Ayers, J. E. Reardon, G. B. Roberts, J. A. Fyfe, M. R. Blum, D. R. Averett, R. E. Dornsife, B. A. Domin, R. Ferone, D. A. Lewis, and T. A. Krenitsky. 1994. 5-Chloro-2′,3′-dideoxy-3′-fluorouridine (935U83), a selective anti-human immunodeficiency virus agent with an improved metabolic and toxicological profile. Antimicrob. Agents Chemother. 38:1590-1603. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

