Intensive induction chemotherapy with C-BOP/BEP for intermediate- and poor-risk metastatic germ cell tumours (EORTC trial 30948)
- PMID: 16251877
- PMCID: PMC2361516
- DOI: 10.1038/sj.bjc.6602830
Intensive induction chemotherapy with C-BOP/BEP for intermediate- and poor-risk metastatic germ cell tumours (EORTC trial 30948)
Abstract
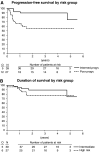
New chemotherapy regimens are continuously explored in patients with high-risk malignant germ cell tumours (MGCTs). This multicentre phase II trial assessed the efficacy and toxicity of C-BOP/BEP chemotherapy in intermediate and poor prognosis MGCT (IGCCCG criteria). C-BOP/BEP treatment consisted of cycles of cisplatin, vincristine, bleomycin and carboplatin, followed by one cycle of vincristine and bleomycin and three cycles of BEP (bleomycon, etoposide, cisplatin). The trial was designed to demonstrate a 1-year progression-free survival rate of 80%, that is, to exclude a 1-year rate of 70% or less, with a one-sided significance level of 5%. Secondary end points included toxicity, overall survival and the postchemotherapy complete response rate. In total, 16 European hospitals entered 66 eligible patients (intermediate prognosis group: 37; poor prognosis group: 29). A total of 45 patients (68.2%, 95% confidence interval (95% CI): 56.9-79.4%) achieved a complete response (intermediate prognosis: 30; poor prognosis: 15). After a median observation time of 40.4 months (range: 13.7-66.3), the 1-year progression-free survival rate was 81.8% 95% CI: 72.5-91.1%). The 2-year overall survival was 84.5% (95% CI: 75.6-93.3%). In all, 51 patients experienced at least one episode of WHO grade 3/4 leucopenia, and at least one event of grade 3/4 thrombocytopenia occurred in 30 patients. There was no toxic death. With an 82% 1-year progression-free survival and a lower limit of the 95% CI above 70%, the efficacy of C-BOP/BEP is comparable to that of published alternative chemotherapy schedules in high-risk MGCT patients. The treatment's toxicity is manageable in a multicentre setting. In poor prognosis patients, C-BOP/BEP should be compared to standard chemotherapy of four cycles of BEP.
Figures
Similar articles
-
Bleomycin, vincristine, cisplatin/bleomycin, etoposide, cisplatin chemotherapy: an alternating, dose intense regimen producing promising results in untreated patients with intermediate or poor prognosis malignant germ-cell tumours.Br J Cancer. 2004 Feb 9;90(3):601-6. doi: 10.1038/sj.bjc.6601528. Br J Cancer. 2004. PMID: 14760371 Free PMC article. Clinical Trial.
-
A randomised phase 2 trial of intensive induction chemotherapy (CBOP/BEP) and standard BEP in poor-prognosis germ cell tumours (MRC TE23, CRUK 05/014, ISRCTN 53643604).Eur Urol. 2015 Mar;67(3):534-43. doi: 10.1016/j.eururo.2014.06.034. Epub 2014 Jul 4. Eur Urol. 2015. PMID: 25001888 Free PMC article. Clinical Trial.
-
Long-term outcomes with intensive induction chemotherapy (carboplatin, bleomycin, vincristine and cisplatin/bleomycin, etoposide and cisplatin) and standard bleomycin, etoposide and cisplatin in poor prognosis germ cell tumours: A randomised phase II trial (ISRCTN53643604).Eur J Cancer. 2020 Mar;127:139-149. doi: 10.1016/j.ejca.2019.12.028. Epub 2020 Jan 31. Eur J Cancer. 2020. PMID: 32007714 Free PMC article. Clinical Trial.
-
Intensive induction chemotherapy with CBOP/BEP in patients with poor prognosis germ cell tumors.J Clin Oncol. 2003 Mar 1;21(5):871-7. doi: 10.1200/JCO.2003.05.155. J Clin Oncol. 2003. PMID: 12610187 Review.
-
Management of good risk germ-cell tumours.BJU Int. 2009 Nov;104(9 Pt B):1387-91. doi: 10.1111/j.1464-410X.2009.08864.x. BJU Int. 2009. PMID: 19840018 Review.
Cited by
-
Prognostic factors and efficacy of different chemotherapeutic regimens in patients with mediastinal nonseminomatous germ cell tumors.J Cancer Res Clin Oncol. 2014 Feb;140(2):311-8. doi: 10.1007/s00432-013-1567-1. Epub 2013 Dec 17. J Cancer Res Clin Oncol. 2014. PMID: 24337455 Free PMC article. Clinical Trial.
-
Standard-Dose Versus High-Dose Cisplatin for Intermediate/Poor-Risk Extracranial Malignant Germ Cell Tumors: Re-Analysis of Pediatric Oncology Group 9049 and Children's Cancer Group 8882 Trial Using Updated MaGIC Risk Stratification.Pediatr Blood Cancer. 2025 Jun;72(6):e31665. doi: 10.1002/pbc.31665. Epub 2025 Mar 17. Pediatr Blood Cancer. 2025. PMID: 40098231
-
Is CBOP/BEP an alternative to BEP for patients with poor prognosis metastatic germ cell tumours?ESMO Open. 2016 Aug 25;1(4):e000089. doi: 10.1136/esmoopen-2016-000089. eCollection 2016. ESMO Open. 2016. PMID: 27843634 Free PMC article. No abstract available.
-
Accelerated BEP: a phase I trial of dose-dense BEP for intermediate and poor prognosis metastatic germ cell tumour.Br J Cancer. 2011 Sep 6;105(6):766-72. doi: 10.1038/bjc.2011.309. Epub 2011 Aug 16. Br J Cancer. 2011. PMID: 21847130 Free PMC article. Clinical Trial.
-
Urologic oncology: Poor trial accrual hinders germ cell tumor therapy advances.Nat Rev Urol. 2012 Apr 3;9(5):243-5. doi: 10.1038/nrurol.2012.59. Nat Rev Urol. 2012. PMID: 22472588 No abstract available.
References
-
- Aass N, Klepp O, Cavallin-Stahl E, Dahl O, Wicklund H, Unsgaard B, Baldetorp L, Ahlstrom S, Fossa SD (1991) Prognostic factors in unselected patients with nonseminomatous metastatic testicular cancer: a multicenter experience. J Clin Oncol 9: 818–826 - PubMed
-
- Anthoney DA, Kaye SB (1999) Treatment of poor prognosis metastatic germ-cell tumours: much heat but, as yet, little light. Ann Oncol 10: 255–258 - PubMed
-
- Bower M, Newlands ES, Holden L, Rustin GJS, Begent RHJ (1997) Treatment of men with metastatic non-seminomatous germ cell tumours with cyclical POMB/ACE chemotherapy. Ann Oncol 8: 477–483 - PubMed
-
- Christian JA, Huddart RA, Norman A, Mason M, Fossa SD, Aass N, Nicholl EJ, Dearnaley DP, Horwich A (2003) Intensive induction chemotherapy with CBOP/BEP in patients with poor prognosis germ cell tumors. J Clin Oncol 21: 871–877 - PubMed
-
- Collette L, Sylvester RJ, Stenning SP, Fossa SD, Mead GM, de Wit R, de Mulder PH, Neymark N, Lallemand E, Kaye SB (1999) Impact of treating institution on survival of patients with ‘poor-prognosis’ metastatic nonseminoma. European Organization for Research and Treatment of Cancer Genito-Urinary Tract Cancer Collaborative Group and the Medical Research Council Testicular Cancer Working Party. J Natl Cancer Inst 91 (10): 839–846 - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical