Minimal features of paxillin that are required for the tyrosine phosphorylation of focal adhesion kinase
- PMID: 16253116
- PMCID: PMC1360707
- DOI: 10.1042/BJ20051241
Minimal features of paxillin that are required for the tyrosine phosphorylation of focal adhesion kinase
Abstract
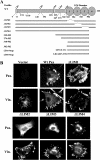
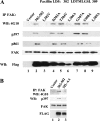
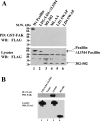
Tyrosine phosphorylation of FAK (focal adhesion kinase) regulates signalling that results from the interaction of integrins with extracellular matrix and growth factor receptors. A critical step in this process is the phosphorylation of Tyr397 of FAK, which creates a binding site for Src family kinases, PI3K (phosphoinositide 3-kinase) and Shc (Src homology and collagen homology). An intact Tyr397 site is required for FAK-mediated regulation of cell migration, survival signals and full responsiveness to soluble growth factors. We showed previously that the adaptor protein paxillin is required for the overall tyrosine phosphorylation of FAK in embryonic stem cells [Wade, Bohl and Vande Pol (2002) Oncogene 21, 96-107]. In the present paper, we identify the minimal structural features of paxillin that are required to support overall FAK tyrosine phosphorylation and Tyr397 phosphorylation. Paxillin contains N-terminal leucine-rich LD motifs that bind directly to FAK and four LIM (Lin-11, Isl-1 and Mec-3) domains in the C-terminus. We show that paxillin LIM domains 1, 2 and 3 are each required for FAK tyrosine phosphorylation, while LIM4 is dispensable. In addition to paxillin LIM domains 1, 2 and 3, a single LD motif on paxillin is required to support FAK tyrosine phosphorylation in embryonic stem cells. Both sequence and spatial requirements exist for LD motifs to support FAK tyrosine phosphorylation. Interestingly, synthetic LD motifs that fail to bind FAK in vitro are able to fully support FAK tyrosine phosphorylation, indicating that minimal interactions of LD motifs with FAK suffice. Our results demonstrate at least four distinct structural domains of paxillin support at least three distinct functions that are each required for FAK tyrosine phosphorylation.
Figures
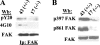



Similar articles
-
Paxillin null embryonic stem cells are impaired in cell spreading and tyrosine phosphorylation of focal adhesion kinase.Oncogene. 2002 Jan 3;21(1):96-107. doi: 10.1038/sj.onc.1205013. Oncogene. 2002. PMID: 11791180
-
Identification of LIM3 as the principal determinant of paxillin focal adhesion localization and characterization of a novel motif on paxillin directing vinculin and focal adhesion kinase binding.J Cell Biol. 1996 Nov;135(4):1109-23. doi: 10.1083/jcb.135.4.1109. J Cell Biol. 1996. PMID: 8922390 Free PMC article.
-
Structural basis for the interaction between focal adhesion kinase and CD4.J Mol Biol. 2008 Feb 1;375(5):1320-8. doi: 10.1016/j.jmb.2007.11.040. Epub 2007 Nov 22. J Mol Biol. 2008. PMID: 18078954
-
Paxillin: a focal adhesion-associated adaptor protein.Oncogene. 2001 Oct 1;20(44):6459-72. doi: 10.1038/sj.onc.1204786. Oncogene. 2001. PMID: 11607845 Review.
-
Focal adhesion kinase: from in vitro studies to functional analyses in vivo.Curr Protein Pept Sci. 2011 Feb;12(1):52-67. doi: 10.2174/138920311795659452. Curr Protein Pept Sci. 2011. PMID: 21190526 Review.
Cited by
-
Human papillomavirus E6 regulates the cytoskeleton dynamics of keratinocytes through targeted degradation of p53.J Virol. 2007 Nov;81(22):12675-9. doi: 10.1128/JVI.01083-07. Epub 2007 Sep 5. J Virol. 2007. PMID: 17804489 Free PMC article.
-
Structural and functional analysis of LIM domain-dependent recruitment of paxillin to αvβ3 integrin-positive focal adhesions.Commun Biol. 2021 Mar 29;4(1):380. doi: 10.1038/s42003-021-01886-9. Commun Biol. 2021. PMID: 33782527 Free PMC article.
-
Paxillin enables attachment-independent tyrosine phosphorylation of focal adhesion kinase and transformation by RAS.J Biol Chem. 2011 Nov 4;286(44):37932-37944. doi: 10.1074/jbc.M111.294504. Epub 2011 Sep 7. J Biol Chem. 2011. PMID: 21900245 Free PMC article.
-
Interactions between E6, FAK, and GIT1 at paxillin LD4 are necessary for transformation by bovine papillomavirus 1 E6.J Virol. 2014 Sep 1;88(17):9927-33. doi: 10.1128/JVI.00552-14. Epub 2014 Jun 18. J Virol. 2014. PMID: 24942580 Free PMC article.
-
The Chlamydia effector TarP mimics the mammalian leucine-aspartic acid motif of paxillin to subvert the focal adhesion kinase during invasion.J Biol Chem. 2014 Oct 31;289(44):30426-30442. doi: 10.1074/jbc.M114.604876. Epub 2014 Sep 5. J Biol Chem. 2014. PMID: 25193659 Free PMC article.
References
-
- Brown N. H. Cell-cell adhesion via the ECM: integrin genetics in fly and worm. Matrix Biol. 2000;19:191–201. - PubMed
-
- Bokel C., Brown N. H. Integrins in development: moving on, responding to, and sticking to the extracellular matrix. Dev. Cell. 2002;3:311–321. - PubMed
-
- Juliano R. L. Signal transduction by cell adhesion receptors and the cytoskeleton: functions of integrins, cadherins, selectins, and immunoglobulin-superfamily members. Annu. Rev. Pharmacol. Toxicol. 2002;42:283–323. - PubMed
-
- Danen E. H., Yamada K. M. Fibronectin, integrins, and growth control. J. Cell. Physiol. 2001;189:1–13. - PubMed
-
- Martin K. H., Slack J. K., Boerner S. A., Martin C. C., Parsons J. T. Integrin connections map: to infinity and beyond. Science. 2002;296:1652–1653. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

