Pathology parameters and adjuvant tamoxifen response in a randomised premenopausal breast cancer trial
- PMID: 16254100
- PMCID: PMC1770762
- DOI: 10.1136/jcp.2005.027185
Pathology parameters and adjuvant tamoxifen response in a randomised premenopausal breast cancer trial
Abstract
Background: Subgroups of breast cancer that have an impaired response to endocrine treatment, despite hormone receptor positivity, are still poorly defined. Breast cancer can be subdivided according to standard pathological parameters including histological type, grade, and assessment of proliferation. These parameters are the net result of combinations of genetic alterations effecting tumour behaviour and could potentially reflect subtypes that respond differently to endocrine treatment.
Aims: To investigate the usefulness of these parameters as predictors of the response to tamoxifen in premenopausal women with breast cancer.
Materials/methods: Clinically established pathological parameters were assessed and related to the tamoxifen response in 500 available tumour specimens from 564 premenopausal patients with breast cancer randomised to either two years of tamoxifen or no treatment with 14 years of follow up. Proliferation was further evaluated by immunohistochemical Ki-67 expression.
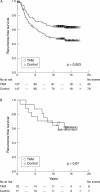
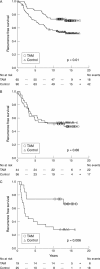
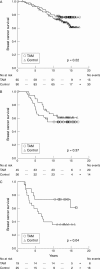
Results: Oestrogen receptor positive ductal carcinomas responded as expected to tamoxifen, whereas the difference in recurrence free survival between control and tamoxifen treated patients was less apparent in the relatively few lobular carcinomas. For histological grade, there was no obvious difference in treatment response between the groups. The relation between proliferation and tamoxifen response seemed to be more complex, with a clear response in tumours with high and low proliferation, whereas tumours with intermediate proliferation defined by Ki-67 responded more poorly.
Conclusions: Clinically established pathology parameters seem to mirror the endocrine treatment response and could potentially be valuable in future treatment decisions for patients with breast cancer.
Figures
Similar articles
-
Prognostic role of human epidermal growth factor receptor 2 status in premenopausal early breast cancer treated with adjuvant tamoxifen.Clin Breast Cancer. 2013 Aug;13(4):247-53. doi: 10.1016/j.clbc.2013.02.005. Epub 2013 May 15. Clin Breast Cancer. 2013. PMID: 23684167
-
[Receptor (ER, PgR) levels as prognostic factors in the endocrine therapy of pre- and post-menopausal patients with stage III infiltrative ductal and lobular cancer of the breast].Ginecol Obstet Mex. 1996 Mar;64:135-9. Ginecol Obstet Mex. 1996. PMID: 8729191 Clinical Trial. Spanish.
-
Outcome prediction for estrogen receptor-positive breast cancer based on postneoadjuvant endocrine therapy tumor characteristics.J Natl Cancer Inst. 2008 Oct 1;100(19):1380-8. doi: 10.1093/jnci/djn309. Epub 2008 Sep 23. J Natl Cancer Inst. 2008. PMID: 18812550 Free PMC article.
-
The nature of tamoxifen action in the control of female breast cancer.In Vivo. 2001 Jul-Aug;15(4):319-25. In Vivo. 2001. PMID: 11695224 Review.
-
The continued evidence from overviews: what is the clinical utility?Breast. 2013 Aug;22 Suppl 2:S8-11. doi: 10.1016/j.breast.2013.07.002. Breast. 2013. PMID: 24074798 Review.
Cited by
-
Can predictive biomarkers in breast cancer guide adjuvant endocrine therapy?Nat Rev Clin Oncol. 2012 Sep;9(9):529-41. doi: 10.1038/nrclinonc.2012.121. Epub 2012 Jul 24. Nat Rev Clin Oncol. 2012. PMID: 22825374 Review.
-
Is Chemoendocrine Treatment without Alternative?Breast Care (Basel). 2008;3(4):231-235. doi: 10.1159/000149558. Epub 2008 Aug 22. Breast Care (Basel). 2008. PMID: 21076602 Free PMC article. No abstract available.
-
Digital Image Analysis of Ki-67 Stained Tissue Microarrays and Recurrence in Tamoxifen-Treated Breast Cancer Patients.Clin Epidemiol. 2020 Jul 20;12:771-781. doi: 10.2147/CLEP.S248167. eCollection 2020. Clin Epidemiol. 2020. PMID: 32801916 Free PMC article.
-
Long Noncoding RNA Signature and Disease Outcome in Estrogen Receptor-Positive Breast Cancer Patients Treated with Tamoxifen.J Breast Cancer. 2018 Sep;21(3):277-287. doi: 10.4048/jbc.2018.21.e39. Epub 2018 Sep 12. J Breast Cancer. 2018. PMID: 30275856 Free PMC article.
-
Tamoxifen therapy benefit for patients with 70-gene signature high and low risk.Breast Cancer Res Treat. 2017 Nov;166(2):593-601. doi: 10.1007/s10549-017-4428-9. Epub 2017 Aug 4. Breast Cancer Res Treat. 2017. PMID: 28776283 Free PMC article.
References
-
- Early Breast Cancer Trialist’s Collaborative Group. Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet 1998;351:1451–67. - PubMed
-
- Ferno M, Baldetorp B, Hatschek T, et al. Results of two or five years of adjuvant tamoxifen correlated to steroid receptor and S-phase levels. Breast Cancer Res Treat 2000;59:69–76. - PubMed
-
- Bryant J, Fisher B, Dignam J. Duration of adjuvant tamoxifen therapy. J Natl Cancer Inst 2001;30:56–61. - PubMed
-
- Stewart H, Prescott R, Forrest P. Scottish adjuvant tamoxifen trial: a randomised study updated to 15 years. J Natl Cancer Inst 2001;93:456–62. - PubMed
-
- Swedish Breast Cancer Cooperative Group. Randomized trial of two versus five years of adjuvant tamoxifen for postmenopausal early stage breast cancer. J Natl Cancer Inst 1996;88:1543–9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
