Inhibition of polyprotein processing and RNA replication of human rhinovirus by pyrrolidine dithiocarbamate involves metal ions
- PMID: 16254325
- PMCID: PMC1280194
- DOI: 10.1128/JVI.79.22.13892-13899.2005
Inhibition of polyprotein processing and RNA replication of human rhinovirus by pyrrolidine dithiocarbamate involves metal ions
Abstract
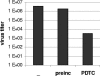
Pyrrolidine dithiocarbamate (PDTC) is an antiviral compound that was shown to inhibit the replication of human rhinoviruses (HRVs), poliovirus, and influenza virus. To elucidate the mechanism of PDTC, the effects on the individual steps of the infection cycle of HRV were investigated. PDTC did not interfere with receptor binding or internalization by receptor mediated endocytosis of HRV2 particles into HeLa cells. But we demonstrate that the processing of the viral polyprotein was prevented by PDTC treatment in HeLa cells infected with HRV2. Furthermore, PDTC inhibited the replication of the viral RNA, even when added four hours post infection. As PDTC is described as a metal ion binding agent, we investigated the effect of other metal chelators on the multiplication of HRV2. We show that EDTA, omicron-phenanthroline, and bathocuproine disulfonic acid do not exhibit any antiviral properties. Surprisingly, these substances, coadministered with PDTC, abolished the antiviral effect of PDTC, suggesting that metal ions play a pivotal role in the inhibition of virus multiplication. These results suggest that PDTC inhibits the activity of the viral proteases in a metal ion dependent way.
Figures



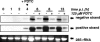


References
-
- Bessho, R., K. Matsubara, M. Kubota, K. Kuwakado, H. Hirota, Y. Wakazono, Y. W. Lin, A. Okuda, M. Kawai, R. Nishikomori, and et al. 1994. Pyrrolidine dithiocarbamate, a potent inhibitor of nuclear factor kappa B (NF-kappa B) activation, prevents apoptosis in human promyelocytic leukemia HL-60 cells and thymocytes. Biochem. Pharmacol. 48:1883-1889. - PubMed
-
- Bonifacino, J. S. (ed.). 1998. Current protocols in cell biology. John Wiley & Sons Inc., New York, NY.
-
- Chonmaitree, T., V. M. Howie, and A. L. Truant. 1986. Presence of respiratory viruses in middle ear fluids and nasal wash specimens from children with acute otitis media. Pediatrics 77:698-702. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

