Immunogenicity of recombinant fiber-chimeric adenovirus serotype 35 vector-based vaccines in mice and rhesus monkeys
- PMID: 16254351
- PMCID: PMC1280229
- DOI: 10.1128/JVI.79.22.14161-14168.2005
Immunogenicity of recombinant fiber-chimeric adenovirus serotype 35 vector-based vaccines in mice and rhesus monkeys
Abstract
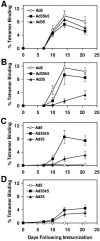
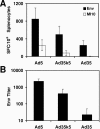
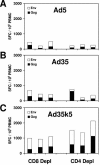
Preexisting immunity to adenovirus serotype 5 (Ad5) has been shown to suppress the immunogenicity of recombinant Ad5 (rAd5) vector-based vaccines for human immunodeficiency virus type 1 (HIV-1) in both preclinical studies and clinical trials. A potential solution to this problem is to utilize rAd vectors derived from rare Ad serotypes, such as Ad35. However, rAd35 vectors have appeared less immunogenic than rAd5 vectors in preclinical studies to date. In this study, we explore the hypothesis that the differences in immunogenicity between rAd5 and rAd35 vectors may be due in part to differences between the fiber proteins of these viruses. We constructed capsid chimeric rAd35 vectors containing the Ad5 fiber knob (rAd35k5) and compared the immunogenicities of rAd5, rAd35k5, and rAd35 vectors expressing simian immunodeficiency virus Gag and HIV-1 Env in mice and rhesus monkeys. In vitro studies demonstrated that rAd35k5 vectors utilized the Ad5 receptor CAR rather than the Ad35 receptor CD46. In vivo studies showed that rAd35k5 vectors were more immunogenic than rAd35 vectors in both mice and rhesus monkeys. These data suggest that the Ad5 fiber knob contributes substantially to the immunogenicity of rAd vectors. Moreover, these studies demonstrate that capsid chimeric rAd vectors can be constructed to combine beneficial immunologic and serologic properties of different Ad serotypes.
Figures





References
-
- Altman, J. D., P. A. H. Moss, P. J. R. Goulder, D. H. Barouch, M. G. McHeyzer-Williams, J. I. Bell, A. J. McMichael, and M. M. Davis. 1996. Phenotypic analysis of antigen-specific T lymphocytes. Science 274:94-96. - PubMed
-
- Barouch, D. H., P. F. McKay, S. M. Sumida, S. Santra, S. S. Jackson, D. A. Gorgone, M. A. Lifton, B. K. Chakrabarti, L. Xu, G. J. Nabel, and N. L. Letvin. 2003. Plasmid chemokines and colony-stimulating factors enhance the immunogenicity of DNA priming-viral vector boosting human immunodeficiency virus type 1 vaccines. J. Virol. 77:8729-8735. - PMC - PubMed
-
- Barouch, D. H., M. G. Pau, J. H. Custers, W. Koudstaal, S. Kostense, M. J. Havenga, D. M. Truitt, S. M. Sumida, M. G. Kishko, J. C. Arthur, B. Korioth-Schmitz, M. H. Newberg, D. A. Gorgone, M. A. Lifton, D. L. Panicali, G. J. Nabel, N. L. Letvin, and J. Goudsmit. 2004. Immunogenicity of recombinant adenovirus serotype 35 vaccine in the presence of pre-existing anti-Ad5 immunity. J. Immunol. 172:6290-6297. - PubMed
-
- Barouch, D. H., J. Powers, D. M. Truitt, M. G. Kishko, J. C. Arthur, F. W. Peyerl, M. J. Kuroda, D. A. Gorgone, M. A. Lifton, C. I. Lord, V. M. Hirsch, D. C. Montefiori, A. Carville, K. G. Mansfield, K. J. Kunstman, S. M. Wolinsky, and N. L. Letvin. 2005. Dynamic immune responses maintain cytotoxic T lymphocyte epitope mutations in transmitted simian immunodeficiency virus variants. Nat. Immunol. 6:247-252. - PubMed
-
- Barouch, D. H., and G. J. Nabel. 2005. Adenovirus vector-based vaccines for human immunodeficiency virus type 1. Hum. Gene Ther. 16:149-156. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

