High affinity acylating antagonists for muscarinic receptors
- PMID: 1625525
- PMCID: PMC3469257
- DOI: 10.1016/0024-3205(92)90586-e
High affinity acylating antagonists for muscarinic receptors
Abstract
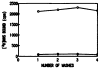
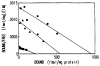
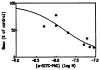
The muscarinic antagonists pirenzepine and telenzepine were derivatized as alkylamino derivatives at a site on the molecules corresponding to a region of bulk tolerance in receptor binding. The distal primary amino groups were coupled to the cross-linking reagent meta-phenylene diisothiocyanate, resulting in two isothiocyanate derivatives that were found to inhibit muscarinic receptors irreversibly and in a dose-dependent fashion. Preincubation of rat forebrain membranes with an isothiocyanate derivative followed by radioligand binding using [3H]N-methylscopolamine diminished the Bmax value, but did not affect the Kd value. The receptor binding site was not restored upon repeated washing, indicating that irreversible inhibition had occurred. IC50 values for the irreversible inhibition at rat forebrain muscarinic receptors were 0.15 nM and 0.19 nM, for derivatives of pirenzepine and telenzepine, respectively. The isothiocyanate derivative of pirenzepine was non-selective as an irreversible muscarinic inhibitor, and the corresponding derivative prepared from telenzepine was 5-fold selective for forebrain (mainly m1) vs. heart (m2) muscarinic receptors.
Figures
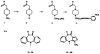

Similar articles
-
Molecular probes for muscarinic receptors: functionalized congeners of selective muscarinic antagonists.Life Sci. 1995;56(11-12):823-30. doi: 10.1016/0024-3205(95)00016-y. Life Sci. 1995. PMID: 10188781 Free PMC article. Review.
-
Molecular probes for muscarinic receptors: derivatives of the M1-antagonist telenzepine.Bioconjug Chem. 1992 May-Jun;3(3):234-40. doi: 10.1021/bc00015a006. Bioconjug Chem. 1992. PMID: 1520727 Free PMC article.
-
Selectivity profile of the novel muscarinic antagonist UH-AH 37 determined by the use of cloned receptors and isolated tissue preparations.Br J Pharmacol. 1991 Jan;102(1):246-50. doi: 10.1111/j.1476-5381.1991.tb12161.x. Br J Pharmacol. 1991. PMID: 2043926 Free PMC article.
-
The binding of [3H]telenzepine to muscarinic acetylcholine receptors in calf forebrain.Eur J Pharmacol. 1988 Jan 5;145(1):87-90. doi: 10.1016/0014-2999(88)90353-6. Eur J Pharmacol. 1988. PMID: 3350039
-
M1 and M3 muscarinic receptor subtypes in rat forebrain.Methods Find Exp Clin Pharmacol. 1991 Dec;13(10):653-60. Methods Find Exp Clin Pharmacol. 1991. PMID: 1770828
Cited by
-
Selective inactivation of muscarinic M2 and M3 receptors in guinea-pig ileum and atria in vitro.Br J Pharmacol. 1993 Aug;109(4):946-52. doi: 10.1111/j.1476-5381.1993.tb13712.x. Br J Pharmacol. 1993. PMID: 8401947 Free PMC article.
-
MUSCARINIC RECEPTOR PROBES BASED ON AMINE CONGENERS OF PIRENZEPINE AND TELENZEPINE.Bioorg Med Chem Lett. 1992 Aug;2(8):845-850. doi: 10.1016/s0960-894x(00)80542-9. Bioorg Med Chem Lett. 1992. PMID: 38213641 Free PMC article.
-
Molecular probes for muscarinic receptors: functionalized congeners of selective muscarinic antagonists.Life Sci. 1995;56(11-12):823-30. doi: 10.1016/0024-3205(95)00016-y. Life Sci. 1995. PMID: 10188781 Free PMC article. Review.
References
-
- TAYLOR P. In: Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 7. Gilman AG, Rall TW, Nies AS, Taylor P, editors. Pergamon Press; New York: 1990. p. 122.
-
- DOODS HN, MATHY MJ, DAVIDESKO D, VAN CHARLDORP KJ, DE JONGE A, VAN WIETEN PA. J Pharmacol Exp Ther. 1987;242:257–262. - PubMed
-
- BONNER TI, BUCKLEY NJ, YOUNG AC, BRANN MR. Science. 1987;237:527–532. - PubMed
-
- BONNER TI, YOUNG AC, BRANN MR, BUCKELY JJ. Neuron. 1988;1:403–410. - PubMed
-
- LIAO CF, THEMMEN AP, JOHO R, BARBERIS C, BIRNBAUMER M, BIRNBAUMER L. J Biol Chem. 1989;264:7328–7337. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

