Intestinal alkaline sphingomyelinase hydrolyses and inactivates platelet-activating factor by a phospholipase C activity
- PMID: 16255717
- PMCID: PMC1386028
- DOI: 10.1042/BJ20051121
Intestinal alkaline sphingomyelinase hydrolyses and inactivates platelet-activating factor by a phospholipase C activity
Abstract
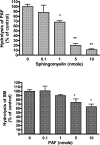
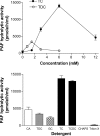
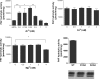
Alkaline sphingomyelinase (alk-SMase) is a new member of the NPP (nucleotide pyrophosphatase/phosphodiesterase) family that hydrolyses SM (sphingomyelin) to generate ceramide in the intestinal tract. The enzyme may protect the intestinal mucosa from inflammation and tumorigenesis. PAF (platelet-activating factor) is a pro-inflammatory phospholipid involved in pathogenesis of inflammatory bowel diseases. We examined whether alk-SMase can hydrolyse and inactivate PAF. [3H]Octadecyl-labelled PAF was incubated with purified rat intestinal alk-SMase or recombinant human alk-SMase expressed in COS-7 cells. The hydrolytic products were assayed with TLC and MS. We found that alkSMase cleaved the phosphocholine head group from PAF and generated 1-O-alkyl-2-acetyl-sn-glycerol. Differing from the activity against SM, the activity against PAF was optimal at pH 7.5, inhibited by EDTA and stimulated by 0.1-0.25 mM Zn2+. The activity was abolished by site mutation of the predicted metal-binding sites that are conserved in all NPP members. Similar to the activity against SM, the activity against PAF was dependent on bile salt, particularly taurocholate and taurochenodeoxycholate. The V(max) for PAF hydrolysis was 374 mumol x h(-1) x (mg of protein)(-1). The hydrolysis of PAF and SM could be inhibited by the presence of SM and PAF respectively, the inhibition of PAF hydrolysis by SM being stronger. The PAF-induced MAPK (mitogen-activated protein kinase) activation and IL-8 (interleukin 8) release in HT-29 cells, and chemotaxis in leucocytes were abolished by alk-SMase treatment. In conclusion, alk-SMase hydrolyses and inactivates PAF by a phospholipase C activity. The finding reveals a novel function, by which alk-SMase may counteract the development of intestinal inflammation and colon cancer.
Figures






Similar articles
-
Identification of human intestinal alkaline sphingomyelinase as a novel ecto-enzyme related to the nucleotide phosphodiesterase family.J Biol Chem. 2003 Oct 3;278(40):38528-36. doi: 10.1074/jbc.M305437200. Epub 2003 Jul 28. J Biol Chem. 2003. PMID: 12885774
-
In vitro effects of fat, FA, and cholesterol on sphingomyelin hydrolysis induced by rat intestinal alkaline sphingomyelinase.Lipids. 2002 May;37(5):469-74. doi: 10.1007/s11745-002-0919-x. Lipids. 2002. PMID: 12056588
-
Purification, characterization, and expression of rat intestinal alkaline sphingomyelinase.J Lipid Res. 2002 Feb;43(2):316-24. J Lipid Res. 2002. PMID: 11861674
-
Alkaline sphingomyelinase: an old enzyme with novel implications.Biochim Biophys Acta. 2006 Mar;1761(3):281-91. doi: 10.1016/j.bbalip.2006.03.007. Epub 2006 Mar 30. Biochim Biophys Acta. 2006. PMID: 16631405 Review.
-
Alkaline sphingomyelinases and ceramidases of the gastrointestinal tract.Chem Phys Lipids. 1999 Nov;102(1-2):97-105. doi: 10.1016/s0009-3084(99)00078-x. Chem Phys Lipids. 1999. PMID: 11001564 Review.
Cited by
-
Deficiency of alkaline SMase enhances dextran sulfate sodium-induced colitis in mice with upregulation of autotaxin.J Lipid Res. 2018 Oct;59(10):1841-1850. doi: 10.1194/jlr.M084285. Epub 2018 Aug 7. J Lipid Res. 2018. PMID: 30087205 Free PMC article.
-
Potential immunosuppressive effects of Escherichia coli O157:H7 experimental infection on the bovine host.BMC Genomics. 2016 Dec 21;17(1):1049. doi: 10.1186/s12864-016-3374-y. BMC Genomics. 2016. PMID: 28003017 Free PMC article.
-
Digestion of Ceramide 2-Aminoethylphosphonate, a Sphingolipid from the Jumbo Flying Squid Dosidicus gigas, in Mice.Lipids. 2017 Apr;52(4):353-362. doi: 10.1007/s11745-017-4239-0. Epub 2017 Feb 27. Lipids. 2017. PMID: 28243820
-
Cellular function and molecular structure of ecto-nucleotidases.Purinergic Signal. 2012 Sep;8(3):437-502. doi: 10.1007/s11302-012-9309-4. Epub 2012 May 4. Purinergic Signal. 2012. PMID: 22555564 Free PMC article. Review.
-
Hits of a high-throughput screen identify the hydrophobic pocket of autotaxin/lysophospholipase D as an inhibitory surface.Mol Pharmacol. 2013 Sep;84(3):415-24. doi: 10.1124/mol.113.087080. Epub 2013 Jun 21. Mol Pharmacol. 2013. PMID: 23793291 Free PMC article.
References
-
- Prescott S. M., Zimmerman G. A., Stafforini D. M., McIntyre T. M. Platelet-activating factor and related lipid mediators. Annu. Rev. Biochem. 2000;69:419–445. - PubMed
-
- Karasawa K., Harada A., Satoh N., Inoue K., Setaka M. Plasma platelet activating factor-acetylhydrolase (PAF-AH) Prog. Lipid Res. 2003;42:93–114. - PubMed
-
- Izzo A. A. PAF and the digestive tract. A review. J. Pharm. Pharmacol. 1996;48:1103–1111. - PubMed
-
- Izumi T., Shimizu T. Platelet-activating factor receptor: gene expression and signal transduction. Biochim. Biophys. Acta. 1995;1259:317–333. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

