The rational design of vaccines
- PMID: 16257375
- PMCID: PMC7108399
- DOI: 10.1016/S1359-6446(05)03600-7
The rational design of vaccines
Abstract
This review provides an insight into the various opportunities for vaccine intervention, analysis of strategies for vaccine development, vaccine ability to modulate immune responses and resultant rational vaccine design. In addition, wider aspects are considered, such as biotechnological advances, advances in immunological understanding and host-pathogen interactions. The key question addressed here is, with all our research and understanding, have we reached a new echelon in vaccine development, that of rational design?
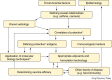
Figures


Similar articles
-
Enhancing vaccine effectiveness with delivery technology.Curr Opin Biotechnol. 2016 Dec;42:24-29. doi: 10.1016/j.copbio.2016.02.022. Epub 2016 Mar 6. Curr Opin Biotechnol. 2016. PMID: 26954947 Free PMC article. Review.
-
A systems framework for vaccine design.Curr Opin Immunol. 2013 Oct;25(5):551-5. doi: 10.1016/j.coi.2013.09.014. Curr Opin Immunol. 2013. PMID: 24358511 Free PMC article. Review.
-
A strategic approach to vaccine development: animal models, monitoring vaccine efficacy, formulation and delivery.Adv Drug Deliv Rev. 2002 Oct 4;54(6):851-61. doi: 10.1016/s0169-409x(02)00072-8. Adv Drug Deliv Rev. 2002. PMID: 12363434 Review.
-
Liposomal vaccine delivery systems.Expert Opin Drug Deliv. 2011 Apr;8(4):505-19. doi: 10.1517/17425247.2011.558081. Epub 2011 Mar 18. Expert Opin Drug Deliv. 2011. PMID: 21413904 Review.
-
Immunological Principles Guiding the Rational Design of Particles for Vaccine Delivery.ACS Nano. 2017 Jan 24;11(1):54-68. doi: 10.1021/acsnano.6b07343. Epub 2017 Jan 11. ACS Nano. 2017. PMID: 28075558 Review.
Cited by
-
Design opportunities for actively targeted nanoparticle vaccines.Nanomedicine (Lond). 2008 Jun;3(3):343-55. doi: 10.2217/17435889.3.3.343. Nanomedicine (Lond). 2008. PMID: 18510429 Free PMC article. Review.
-
Precision Vaccinology Approaches for the Development of Adjuvanted Vaccines Targeted to Distinct Vulnerable Populations.Pharmaceutics. 2023 Jun 19;15(6):1766. doi: 10.3390/pharmaceutics15061766. Pharmaceutics. 2023. PMID: 37376214 Free PMC article. Review.
-
Structure-Based Reverse Vaccinology Failed in the Case of HIV Because it Disregarded Accepted Immunological Theory.Int J Mol Sci. 2016 Sep 21;17(9):1591. doi: 10.3390/ijms17091591. Int J Mol Sci. 2016. PMID: 27657055 Free PMC article. Review.
-
Is it time to switch to a formulation other than the live attenuated poliovirus vaccine to prevent poliomyelitis?Front Public Health. 2024 Jan 8;11:1284337. doi: 10.3389/fpubh.2023.1284337. eCollection 2023. Front Public Health. 2024. PMID: 38259741 Free PMC article. Review.
-
Basic research in HIV vaccinology is hampered by reductionist thinking.Front Immunol. 2012 Jul 9;3:194. doi: 10.3389/fimmu.2012.00194. eCollection 2012. Front Immunol. 2012. PMID: 22787464 Free PMC article.
References
-
- Dentico P. Anamnestic response to administration of purified non-adsorbed hepatitis B surface antigen in healthy responders to hepatitis B vaccine with long-term non-protective antibody titres. Vaccine. 2002;20:3725–3730. - PubMed
-
- Schmitt H.J. The safety, reactogenicity and immunogenicity of a 7-valent pneumococcal conjugate vaccine (7VPnC) concurrently administered with a combination DTaP-IPV-Hib vaccine. Vaccine. 2003;21:3653–3662. - PubMed
-
- Ritvo P. Vaccines in the public eye. Nat. Med. 2005;11(suppl 4):S20–S24. - PubMed
-
- Osterholm M.T. Preparing for the Next Pandemic. N. Engl. J. Med. 2005;352:1839–1842. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

