Serial monitoring of circulating melanoma cells during neoadjuvant biochemotherapy for stage III melanoma: outcome prediction in a multicenter trial
- PMID: 16258104
- PMCID: PMC2856446
- DOI: 10.1200/JCO.2005.02.0958
Serial monitoring of circulating melanoma cells during neoadjuvant biochemotherapy for stage III melanoma: outcome prediction in a multicenter trial
Abstract
Purpose: Circulating tumor cells (CTCs) in blood may be important in assessing tumor progression and treatment response. We hypothesized that quantitative real-time reverse transcriptase polymerase chain reaction using multimarker mRNA assays could detect CTCs and be used as a surrogate predictor of outcome in patients receiving neoadjuvant biochemotherapy (BC) for melanoma.
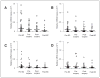
Patients and methods: Blood specimens were collected at four sampling points from 63 patients enrolled on a prospective multicenter phase II trial of BC before and after surgical treatment of American Joint Committee on Cancer stage III melanoma. Each specimen was assessed by quantitative real-time reverse transcriptase polymerase chain reaction for expression of four melanoma-associated markers: melanoma antigen recognized by T cells 1; beta1 --> 4-N-acetylgalactosaminyltransferase; paired box homeotic gene transcription factor 3; and melanoma antigen gene-A3 family, and the changes of CTCs during treatment and prognostic effect of CTCs after overall treatment on recurrence and survival were investigated.
Results: At a median postoperative follow-up time of 30.4 months, 44 (70%) patients were clinically disease free. In relapse-free patients, the number of detected markers significantly decreased during preoperative BC (P = .036), during postoperative BC (P = .002), and during overall treatment (P < .0001). Marker detection after overall treatment was associated with significant decreases in relapse-free and overall survival (P < .0001). By multivariate analysis using a Cox proportional-hazards model, the number of markers detected after overall treatment was a significant independent prognostic factor for overall survival (risk ratio, 12.6; 95% CI, 3.16 to 50.5; P = .0003).
Conclusion: Serial monitoring of CTCs in blood may be useful for indicating systemic subclinical disease and predicting outcome of patients receiving neoadjuvant BC for metastatic melanoma.
Conflict of interest statement
Although all authors completed the disclosure declaration, the following authors or their immediate family members indicated a financial interest. No conflict exists for drugs or devices used in a study if they are not being evaluated as part of the investigation. For a detailed description of the disclosure categories, or for more information about ASCO’s conflict of interest policy, please refer to the Author Disclosure Declaration and the Disclosures of Potential Conflicts of Interest section in Information for Contributors.
[Table: see text]
Figures


References
-
- Balch CM, Buzaid AC, Soong SJ, et al. Final version of the American Joint Committee on Cancer staging system for cutaneous melanoma. J Clin Oncol. 2001;19:3635–3648. - PubMed
-
- Greene FL, Page DL, Fleming ID, et al., editors. AJCC Cancer Staging Manual. 6. New York, NY: Springer-Verlag; 2002.
-
- O’Day SJ, Gammon G, Boasberg PD, et al. Advantages of concurrent biochemotherapy modified by decrescendo interleukin-2, granulocyte colony-stimulating factor, and tamoxifen for patients with metastatic melanoma. J Clin Oncol. 1999;17:2752–2761. - PubMed
-
- Richards JM, Gale D, Mehta N, et al. Combination of chemotherapy with interleukin-2 and interferon alfa for the treatment of meta-static melanoma. J Clin Oncol. 1999;17:651–657. - PubMed
-
- Gibbs P, Iannucci A, Becker M, et al. A phase II study of biochemotherapy for the treatment of metastatic malignant melanoma. Melanoma Res. 2000;10:171–179. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

