Phosphorylation by Rho kinase regulates CRMP-2 activity in growth cones
- PMID: 16260611
- PMCID: PMC1280267
- DOI: 10.1128/MCB.25.22.9973-9984.2005
Phosphorylation by Rho kinase regulates CRMP-2 activity in growth cones
Abstract
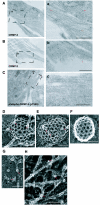
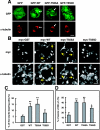
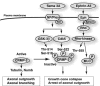
Collapsin response mediator protein 2 (CRMP-2) enhances the advance of growth cones by regulating microtubule assembly and Numb-mediated endocytosis. We previously showed that Rho kinase phosphorylates CRMP-2 during growth cone collapse; however, the roles of phosphorylated CRMP-2 in growth cone collapse remain to be clarified. Here, we report that CRMP-2 phosphorylation by Rho kinase cancels the binding activity to the tubulin dimer, microtubules, or Numb. CRMP-2 binds to actin, but its binding is not affected by phosphorylation. Electron microscopy revealed that CRMP-2 localizes on microtubules, clathrin-coated pits, and actin filaments in dorsal root ganglion neuron growth cones, while phosphorylated CRMP-2 localizes only on actin filaments. The phosphomimic mutant of CRMP-2 has a weakened ability to enhance neurite elongation. Furthermore, ephrin-A5 induces phosphorylation of CRMP-2 via Rho kinase during growth cone collapse. Taken together, these results suggest that Rho kinase phosphorylates CRMP-2, and inactivates the ability of CRMP-2 to promote microtubule assembly and Numb-mediated endocytosis, during growth cone collapse.
Figures




References
-
- Amano, M., K. Chihara, N. Nakamura, Y. Fukata, T. Yano, M. Shibata, M. Ikebe, and K. Kaibuchi. 1998. Myosin II activation promotes neurite retraction during the action of Rho and Rho-kinase. Genes Cells 3:177-188. - PubMed
-
- Amano, M., M. Ito, K. Kimura, Y. Fukata, K. Chihara, T. Nakano, Y. Matsuura, and K. Kaibuchi. 1996. Phosphorylation and activation of myosin by Rho-associated kinase (Rho-kinase). J. Biol. Chem. 271:20246-20249. - PubMed
-
- Amano, M., T. Kaneko, A. Maeda, M. Nakayama, M. Ito, T. Yamauchi, H. Goto, Y. Fukata, N. Oshiro, A. Shinohara, A. Iwamatsu, and K. Kaibuchi. 2003. Identification of Tau and MAP2 as novel substrates of Rho-kinase and myosin phosphatase. J. Neurochem. 87:780-790. - PubMed
-
- Amano, M., Y. Fukata, and K. Kaibuchi. 2000. Regulation and functions of Rho-associated kinase. Exp. Cell Res. 261:44-51. - PubMed
-
- Arimura, N., N. Inagaki, K. Chihara, C. Menager, N. Nakamura, M. Amano, A. Iwamatsu, Y. Goshima, and K. Kaibuchi. 2000. Phosphorylation of collapsin response mediator protein-2 by Rho-kinase. Evidence for two separate signaling pathways for growth cone collapse. J. Biol. Chem. 275:23973-23980. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
