An intracellular antibody can suppress tumorigenicity in hepatitis B virus X-expressing cells
- PMID: 16273352
- PMCID: PMC11030035
- DOI: 10.1007/s00262-005-0037-2
An intracellular antibody can suppress tumorigenicity in hepatitis B virus X-expressing cells
Abstract
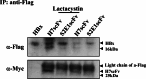
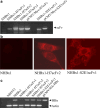
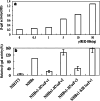
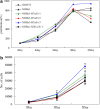
Although the hepatitis B virus X protein (HBx) is thought to play a causative role in the development of hepatocellular carcinoma, it is not yet known whether interfering with HBx function may affect the cellular transformation of HBx-expressing tumor cells. To address this question, we adopted an intracellular antibody fragment expression approach to block the function of HBx. Expression of a single-chain variable fragment (scFv) specific to HBx (designated as H7scFv) inhibited HBx-dependent cellular transactivation. Furthermore, H7scFv suppressed the growth of HBx-expressing tumor cells in both soft agar and nude mice. The suppressive effect of H7scFv on tumorigenicity appeared not to be mediated by inhibition of HBx-induced growth stimulation since the growth rate of these cells was not affected significantly by H7scFv expression. In conclusion, these data suggest that the HBx-dependent transformed phenotype is reversible and that HBx may be a good molecular target for the treatment of HBV-related tumors.
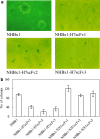
Figures


Similar articles
-
Pin1 interacts with a specific serine-proline motif of hepatitis B virus X-protein to enhance hepatocarcinogenesis.Gastroenterology. 2007 Mar;132(3):1088-103. doi: 10.1053/j.gastro.2006.12.030. Epub 2006 Dec 19. Gastroenterology. 2007. PMID: 17383430
-
HBx protein-mediated ATOH1 downregulation suppresses ARID2 expression and promotes hepatocellular carcinoma.Cancer Sci. 2017 Jul;108(7):1328-1337. doi: 10.1111/cas.13277. Epub 2017 Jun 14. Cancer Sci. 2017. PMID: 28498550 Free PMC article.
-
Hepatitis B virus X protein promotes hepatocellular carcinoma transformation through interleukin-6 activation of microRNA-21 expression.Eur J Cancer. 2014 Oct;50(15):2560-9. doi: 10.1016/j.ejca.2014.07.008. Epub 2014 Jul 30. Eur J Cancer. 2014. PMID: 25087183
-
Hepatitis B virus, HBx mutants and their role in hepatocellular carcinoma.World J Gastroenterol. 2014 Aug 14;20(30):10238-48. doi: 10.3748/wjg.v20.i30.10238. World J Gastroenterol. 2014. PMID: 25132741 Free PMC article. Review.
-
Hepatitis B virus X protein sensitizes UV-induced apoptosis by transcriptional transactivation of Fas ligand gene expression.IUBMB Life. 2005 Sep;57(9):651-8. doi: 10.1080/15216540500239697. IUBMB Life. 2005. PMID: 16203685 Review.
Cited by
-
Intracellular antibody fragment against hepatitis B virus X protein does not inhibit viral replication.Yonsei Med J. 2006 Oct 31;47(5):721-8. doi: 10.3349/ymj.2006.47.5.721. Yonsei Med J. 2006. PMID: 17066517 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

