Safety and efficacy of flecainide in subjects with Long QT-3 syndrome (DeltaKPQ mutation): a randomized, double-blind, placebo-controlled clinical trial
- PMID: 16274417
- PMCID: PMC6932190
- DOI: 10.1111/j.1542-474X.2005.00077.x
Safety and efficacy of flecainide in subjects with Long QT-3 syndrome (DeltaKPQ mutation): a randomized, double-blind, placebo-controlled clinical trial
Abstract
Background: We conducted a study of chronic therapy with flecainide versus placebo in a small group of LQT-3 patients with the DeltaKPQ deletion to evaluate the safety and efficacy of flecainide in this genetic disorder. In vitro studies have shown that flecainide provides correction of the impaired inactivation associated with the DeltaKPQ deletion.
Methods: A randomized, double-blind, placebo-controlled clinical trial was conducted with flecainide and placebo in six male LQT-3 subjects with the DeltaKPQ deletion.
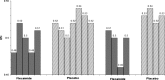
Results: The lowest possible dose of flecainide associated with at least a 40 ms reduction in the QTc interval was determined in an initial open-label, dose-ranging investigation using one-fourth or half of the recommended maximal antiarrhythmic flecainide dose. QTc reduction was achieved with a flecainide dose of 1.5 mg/kg per day in 4 subjects and with 3.0 mg/kg per day in 2 subjects. Subjects were randomized to four 6-month alternating periods of flecainide and placebo therapy based on the open-label dose findings. Average QTc values during placebo and flecainide therapies were 534 ms and 503 ms, respectively, with an adjusted reduction in QTc of -27.1 ms (95% confidence interval: -36.8 ms to -17.4 ms; P<0.001) at a mean flecainide blood level of 0.11+/-0.05 microg/ml. Minimal prolongation in QRS occurred (mean: +2.5 ms), and there were no major adverse cardiac effects.
Conclusions: Chronic low-dose flecainide significantly shortens the QTc interval in LQT-3 subjects with the DeltaKPQ mutation. No major adverse drug effects were observed with flecainide during this trial, but the sample size is not large enough to evaluate the safety of flecainide therapy in patients with this mutation.
Figures


References
-
- Moss AJ, Schwartz PJ, Crampton RS, et al The long QT syndrome. Prospective longitudinal study of 328 families. Circulation 1991;84: 1136–1144. - PubMed
-
- Splawski I, Shen J, Timothy KW, et al Spectrum of mutations in long‐QT syndrome genes. KVLQT1, HERG, SCN5A, KCNE1, and KCNE2. Circulation 2000;102: 1178–1185. - PubMed
-
- Plaster NM, Tawil R, Tristani‐Firouzi M, et al Mutations in Kir2.1 cause the developmental and episodic electrical phenotypes of Andersen's syndrome. Cell 2001;105: 511–519. - PubMed
-
- Mohler PJ, Schott JJ, Gramolini AO, et al Ankyrin‐B mutation causes type 4 long‐QT cardiac arrhythmia and sudden cardiac death. Nature 2003;421: 634–639. - PubMed
-
- Dumaine R, Wang Q, Keating MT, et al Multiple mechanisms of Na+ channel‐linked long‐QT syndrome. Circ Res 1996;78: 916–924. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

