Stress-activated protein kinase pathway functions to support protein synthesis and translational adaptation in response to environmental stress in fission yeast
- PMID: 16278445
- PMCID: PMC1287851
- DOI: 10.1128/EC.4.11.1785-1793.2005
Stress-activated protein kinase pathway functions to support protein synthesis and translational adaptation in response to environmental stress in fission yeast
Abstract
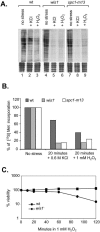
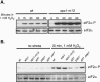
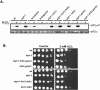
The stress-activated protein kinase (SAPK) pathway plays a central role in coordinating gene expression in response to diverse environmental stress stimuli. We examined the role of this pathway in the translational response to stress in Schizosaccharomyces pombe. Exposing wild-type cells to osmotic stress (KCl) resulted in a rapid but transient reduction in protein synthesis. Protein synthesis was further reduced in mutants disrupting the SAPK pathway, including the mitogen-activated protein kinase Wis1 or the mitogen-activated protein kinase Spc1/Sty1, suggesting a role for these stress response factors in this translational control. Further polysome analyses revealed a role for Spc1 in supporting translation initiation during osmotic stress, and additionally in facilitating translational adaptation. Exposure to oxidative stress (H2O2) resulted in a striking reduction in translation initiation in wild-type cells, which was further reduced in spc1- cells. Reduced translation initiation correlated with phosphorylation of the alpha subunit of eukaryotic initiation factor 2 (eIF2alpha) in wild-type cells. Disruption of Wis1 or Spc1 kinase or the downstream bZip transcription factors Atf1 and Pap1 resulted in a marked increase in eIF2alpha phosphorylation which was dependent on the eIF2alpha kinases Hri2 and Gcn2. These findings suggest a role for the SAPK pathway in supporting translation initiation and facilitating adaptation to environmental stress in part through reducing eIF2alpha phosphorylation in fission yeast.
Figures


References
-
- Dever, T. E. 2002. Gene-specific regulation by general translation factors. Cell 108:545-556. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

