Growth factor-induced shedding of syndecan-1 confers glypican-1 dependence on mitogenic responses of cancer cells
- PMID: 16286510
- PMCID: PMC2171561
- DOI: 10.1083/jcb.200508010
Growth factor-induced shedding of syndecan-1 confers glypican-1 dependence on mitogenic responses of cancer cells
Abstract
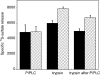
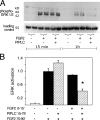
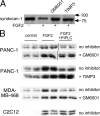
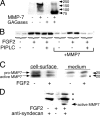
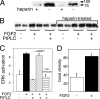
The cell surface heparan sulfate proteoglycan (HSPG) glypican-1 is up-regulated by pancreatic and breast cancer cells, and its removal renders such cells insensitive to many growth factors. We sought to explain why the cell surface HSPG syndecan-1, which is also up-regulated by these cells and is a known growth factor coreceptor, does not compensate for glypican-1 loss. We show that the initial responses of these cells to the growth factor FGF2 are not glypican dependent, but they become so over time as FGF2 induces shedding of syndecan-1. Manipulations that retain syndecan-1 on the cell surface make long-term FGF2 responses glypican independent, whereas those that trigger syndecan-1 shedding make initial FGF2 responses glypican dependent. We further show that syndecan-1 shedding is mediated by matrix metalloproteinase-7 (MMP7), which, being anchored to cells by HSPGs, also causes its own release in a complex with syndecan-1 ectodomains. These results support a specific role for shed syndecan-1 or MMP7-syndecan-1 complexes in tumor progression and add to accumulating evidence that syndecans and glypicans have nonequivalent functions in vivo.
Figures


References
-
- Alexander, C.M., F. Reichsman, M.T. Hinkes, J. Lincecum, K.A. Becker, S. Cumberledge, and M. Bernfield. 2000. Syndecan-1 is required for Wnt-1-induced mammary tumorigenesis in mice. Nat. Genet. 25:329–332. - PubMed
-
- Asundi, V.K., R. Erdman, R.C. Stahl, and D.J. Carey. 2003. Matrix metalloproteinase-dependent shedding of syndecan-3, a transmembrane heparan sulfate proteoglycan, in Schwann cells. J. Neurosci. Res. 73:593–602. - PubMed
-
- Barbareschi, M., P. Maisonneuve, D. Aldovini, M.G. Cangi, L. Pecciarini, F. Angelo Mauri, S. Veronese, O. Caffo, A. Lucenti, P.D. Palma, et al. 2003. High syndecan-1 expression in breast carcinoma is related to an aggressive phenotype and to poorer prognosis. Cancer. 98:474–483. - PubMed
-
- Bellaiche, Y., I. The, and N. Perrimon. 1998. Tout-velu is a Drosophila homologue of the putative tumour suppressor EXT-1 and is needed for Hh diffusion. Nature. 394:85–88. - PubMed

